Basic Information
-
Targeting Strategy

-
Gene targeting strategy for B-hCTLA4/hLAG3 mice. The exon 2 of mouse Ctla4 gene that encodes the extracellular domain was replaced by human CTLA4 exon 2 in B-hCTLA4/hLAG3 mice. The exons 2-3 of mouse Lag3 gene that encode the extracellular domain were replaced by human LAG3 exons 2-3 in B-hCTLA4/hLAG3 mice.
-
Detail

-
Phenotype
Protein Expression Analysis
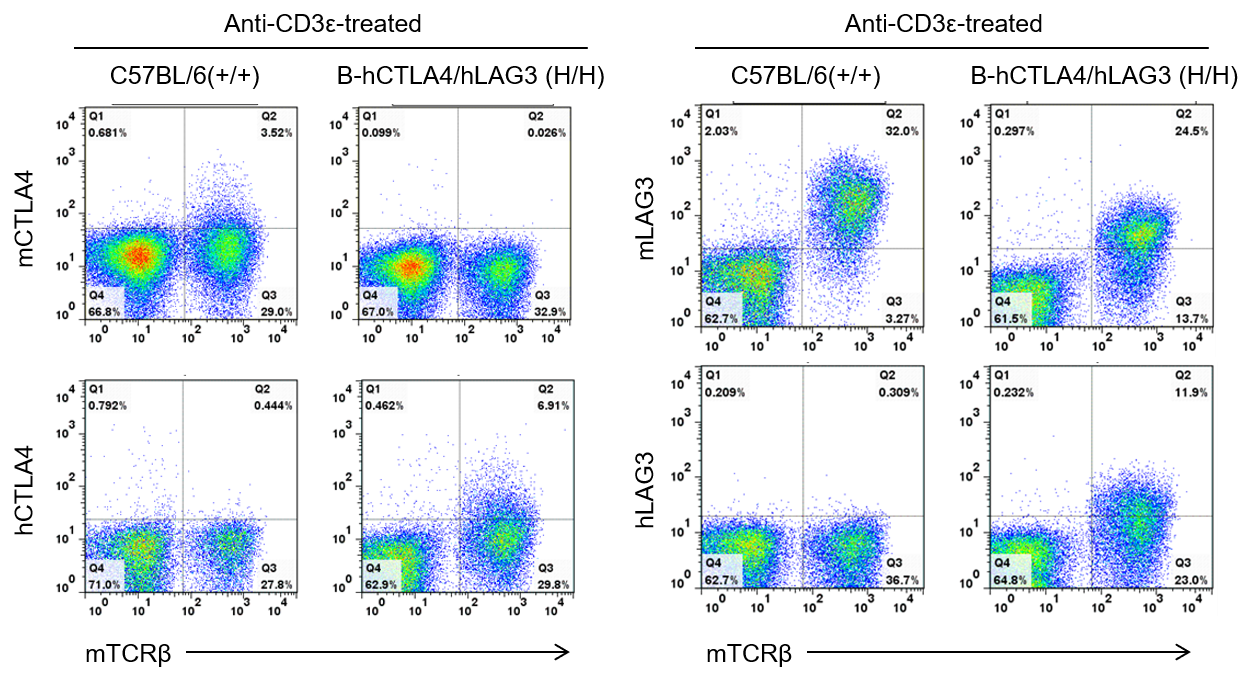
Strain specific CTLA4 and LAG3 expression analysis in homozygous B-hCTLA4/hLAG3 mice by flow cytometry. Splenocytes were collected from WT and homozygous B-hCTLA4/hLAG3 (H/H) mice stimulated with anti-CD3ε in vivo, and analyzed by flow cytometry with species-specific anti-CTLA4 and anti-LAG3 antibody. Mouse CTLA4 was detectable in WT mice, mouse LAG3 was detectable in homozygous B-hCTLA4/hLAG3 and WT mice due to the anti-mouse LAG3 antibody cross-reacts with human LAG3. Human CTLA4 and LAG3 were exclusively detectable in homozygous B-hCTLA4/hLAG3 but not WT mice.
-
References

-
- Cell Research (2018) 0:1–15; doi: 10.1038/s41422-018-0012-z
- Cancer Immunity (22 January 2013) Vol. 13, p. 5
- Journal of Immunological Methods, 18 November 2016. doi: 10.1016/j.jim.2017.04.006


