Basic Information
-
Target strategy

-
This triple knock-in model was developed by breeding B-hPD-1/hPD-L1 mice and the B-hC5AR1 mice together.
Exon 2 of the mouse PD-1 gene that encodes the extracellular domain was replaced by human PD-1 exon 2 in B-hPD-1/hPD-L1/hC5AR1 mice. Exon 3 of the mouse Pdl1 gene that encodes the extracellular domain was replaced by human PD-L1 exon 3 in B-hPD-1/hPD-L1/hC5AR1 mice. Exon 2 of the mouse C5ar1 gene that encodes the full-length protein was replaced by human C5AR1 exon 2 in B-hPD-1/hPD-L1/hC5AR1 mice.
-
Protein expression analysis

-
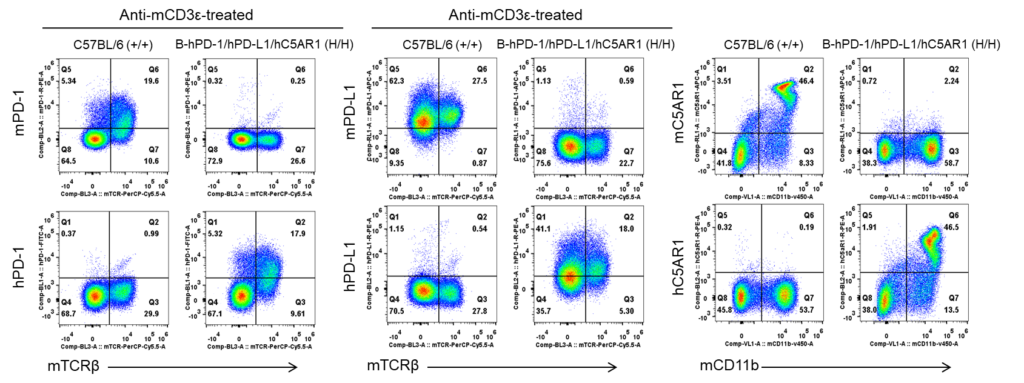
Species-specific PD-1, PD-L1, C5AR1 protein expression analysis in humanized B-hPD-1/hPD-L1/hC5AR1 mice. Splenocytes were isolated from wild-type C57BL/6 (+/+) and homozygous B-hPD-1/hPD-L1/hC5AR1 (H/H) mice and analyzed by flow cytometry using species-specific anti-PD-1 and anti-PD-L1 antibodies. Bone marrow was isolated from wild-type C57BL/6 (+/+) and homozygous B-hPD-1/hPD-L1/hC5AR1 (H/H) mice and analyzed by flow cytometry using species-specific anti-C5AR1 antibodies. Murine PD-1, PD-L1, C5AR1 proteins were detected in wild-type mice, while human PD-1, PD-L1, C5AR1 proteins were exclusively detected in B-hPD-1/hPD-L1/hC5AR1 mice.
-
Analysis of immune cells

-
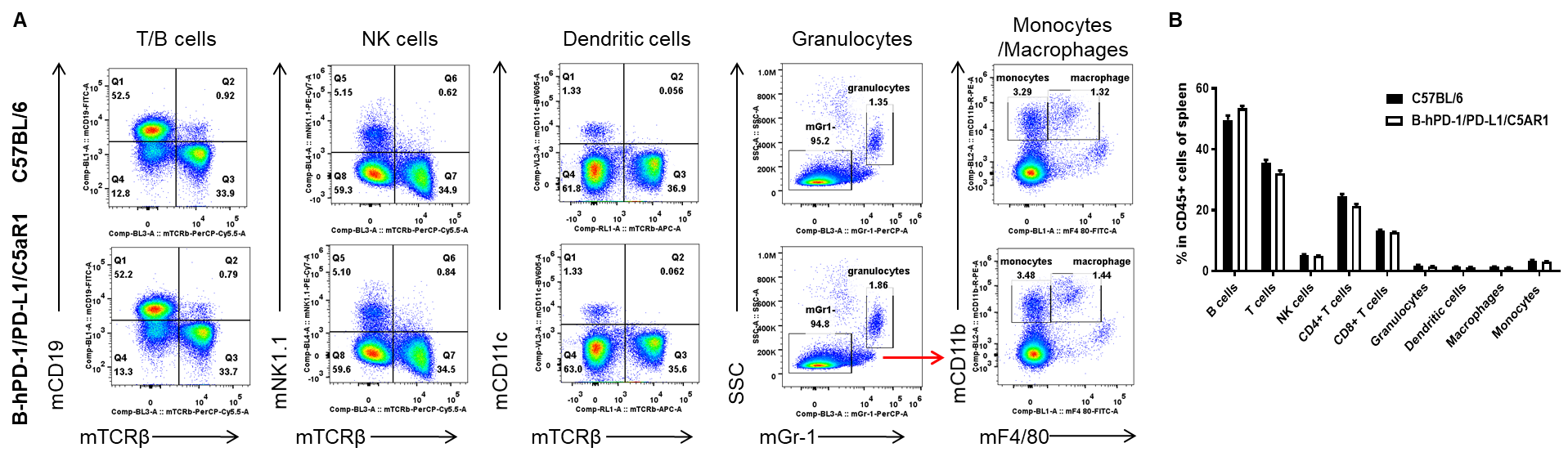
Analysis of spleen leukocyte subpopulations by flow cytometry. Splenocytes were isolated from wild-type C57BL/6 and humanized B-hPD-1/hPD-L1/hC5AR1 mice (female, n=3, 6-week-old), and analyzed by flow cytometry to assess leukocyte subpopulations. (A) Representative flow cytometry plots. Single live cells were gated on CD45+ and used for further analysis as indicated. (B) Percent of T cells, B cells, NK cells, dendritic cells, granulocytes, monocytes and macrophages in B-hPD-1/hPD-L1/hC5AR1 mice were similar to those in wild-type mice, demonstrating that hPD-1, hPD-L1 and hC5AR1 in place of their murine counterparts does not change the overall development, differentiation or distribution of these spleen cell subtypes. Values are expressed as mean ± SEM.
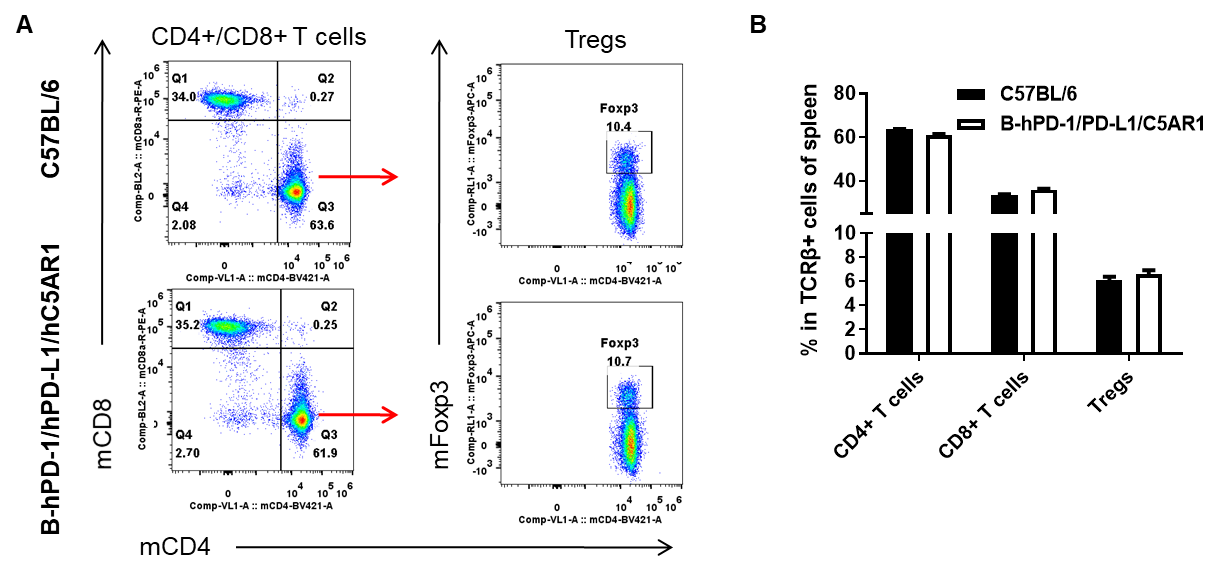
Analysis of spleen T cell subpopulations by flow cytometry. Splenocytes were isolated from wild-type C57BL/6 and humanized B-hPD-1/hPD-L1/hC5AR1 mice (female, n=3, 6-week-old), and analyzed by flow cytometry to assess leukocyte subpopulations. (A) Representative flow cytometry plots. Single live CD45+ cells were gated on TCRβ+ T cells and used for further analysis as indicated. (B) Percent of CD4+ T cells, CD8+ T cells and Tregs in B-hPD-1/hPD-L1/hC5AR1 mice were similar to those in wild-type mice, demonstrating that introduction of hPD-1, hPD-L1 and hC5AR1 in place of their murine counterparts does not change the overall development, differentiation or distribution of these spleen T cell subtypes. Values are expressed as mean ± SEM.
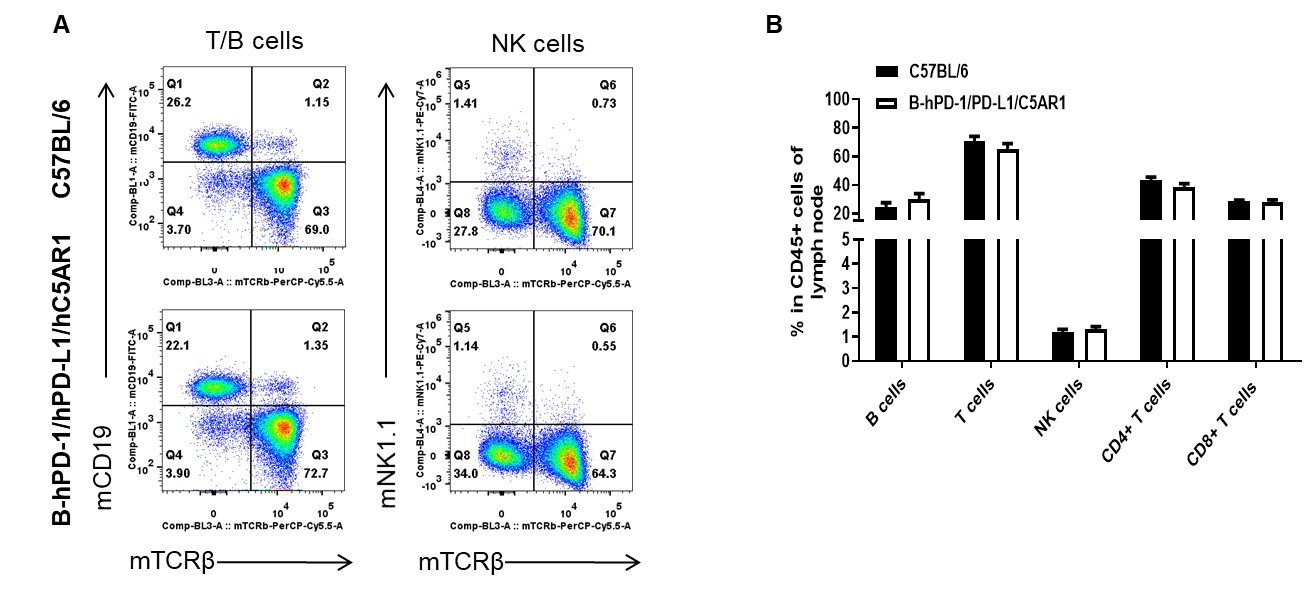
Analysis of lymph node leukocyte subpopulations by FACS. Lymph nodes were isolated from female C57BL/6 and B-hPD-1/hPD-L1/hC5AR1 mice (n=3, 6-week-old). Flow cytometry analysis of the splenocytes was performed to assess leukocyte subpopulations. A. Representative FACS plots. Single live cells were gated for the CD45+ population and used for further analysis as indicated here. B. Results of FACS analysis. Percent of T cells, B cells, NK cells, dendritic cells, granulocytes, monocytes and macrophages in homozygous B-hPD-1/hPD-L1/hC5AR1 mice were similar to those in the C57BL/6 mice, demonstrating that PD-1, PD-L1 and C5AR1 humanized does not change the overall development, differentiation or distribution of these cell types in lymph node. Values are expressed as mean ± SEM.
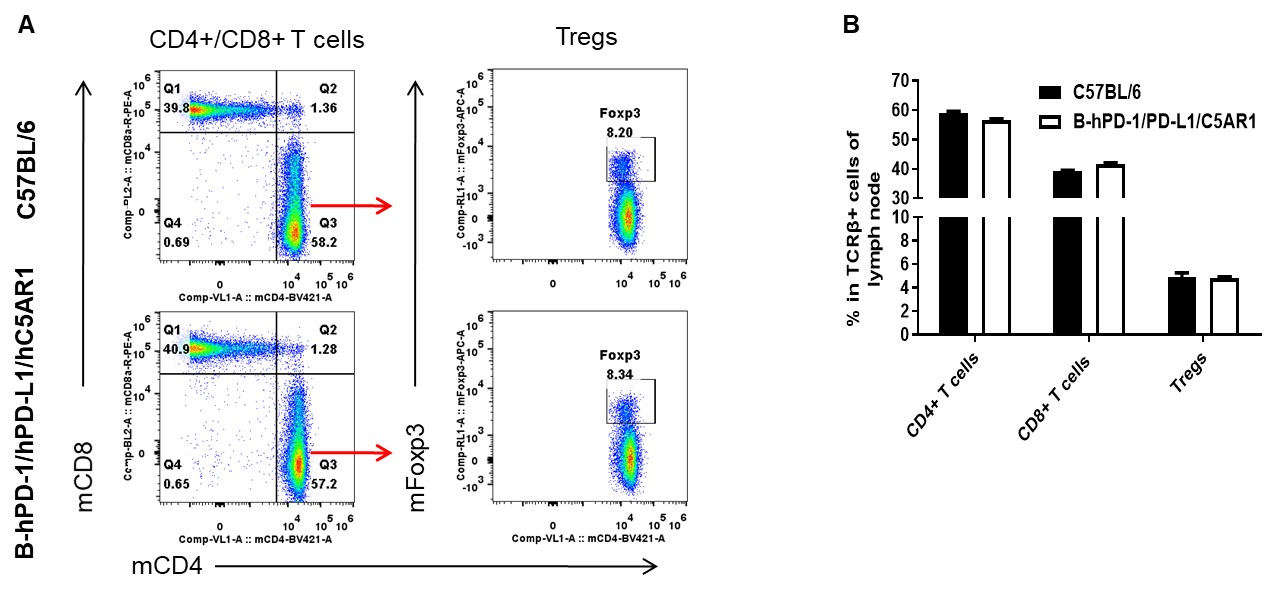
Analysis of lymph node T cell subpopulations by FACS. Leukocytes were isolated from female C57BL/6 and B-hPD-1/hPD-L1/hC5AR1 mice (n=3, 6-week-old). Flow cytometry analysis of the leukocytes was performed to assess leukocyte subpopulations. A. Representative FACS plots. Single live CD45+ cells were gated for TCRβ+ T cell population and used for further analysis as indicated here. B. Results of FACS analysis. The percent of CD4+ T cells, CD8+ T cells, and Tregs in homozygous B-hPD-1/hPD-L1/hC5AR1 mice were similar to those in the C57BL/6 mice, demonstrating that introduction of hPD-1, PD-L1 and C5AR1 in place of its mouse counterpart does not change the overall development, differentiation or distribution of these T cell subtypes in lymph node. Values are expressed as mean ± SEM.
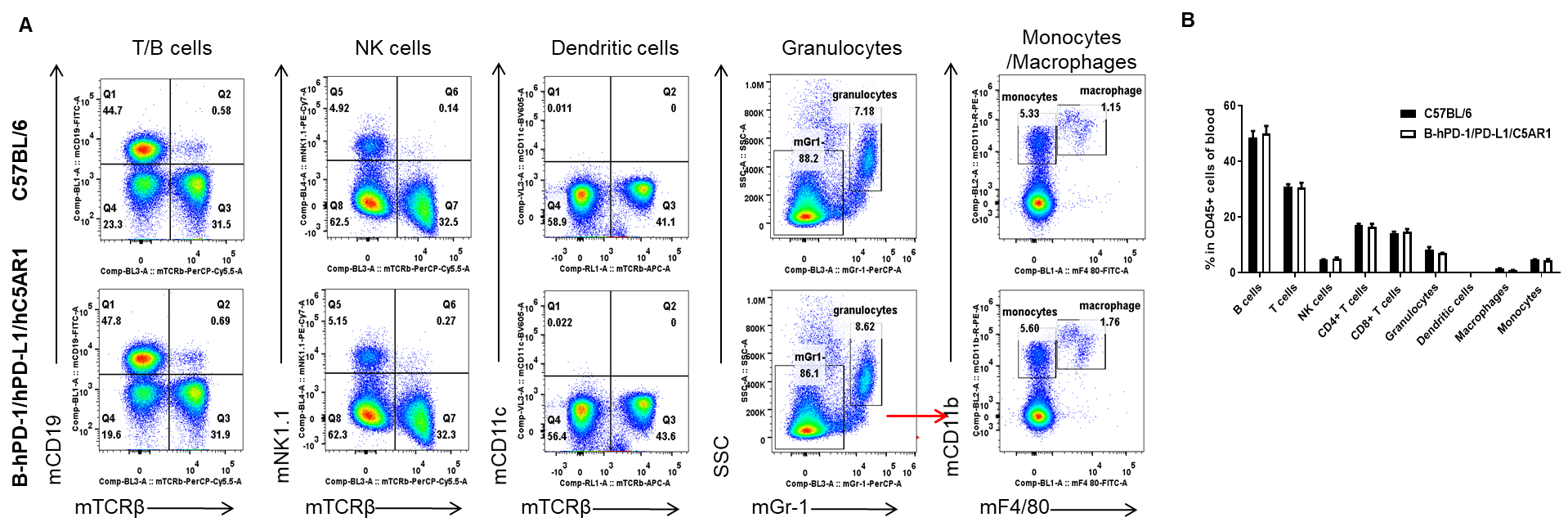
Analysis of blood leukocyte subpopulations by flow cytometry. Blood cells were isolated from wild-type C57BL/6 and humanized B-hPD-1/hPD-L1/hC5AR1 mice (female, n=3, 6-week-old), and analyzed by flow cytometry to assess leukocyte subpopulations. (A) Representative flow cytometry plots. Single live cells were gated on CD45+ and used for further analysis as indicated. (B) Percent of T cells, B cells, NK cells, dendritic cells, granulocytes, monocytes and macrophages in B-hPD-1/hPD-L1/hC5AR1 mice were similar to those in wild-type mice, demonstrating that hPD-1, hPD-L1 and hC5AR1 in place of their murine counterparts does not change the overall development, differentiation or distribution of these blood cell subtypes. Values are expressed as mean ± SEM.
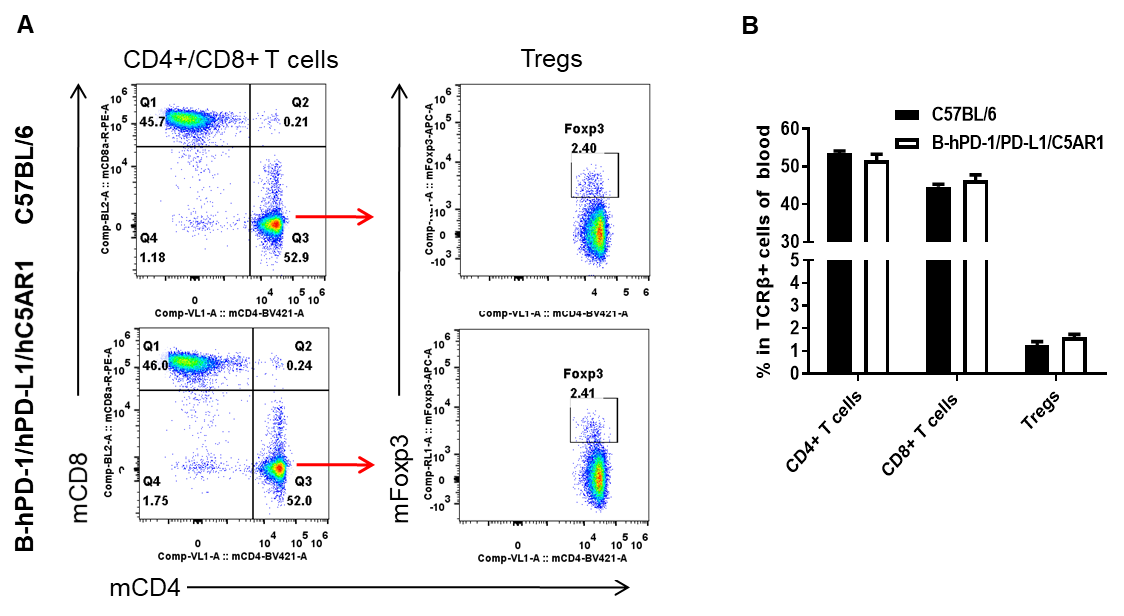
Analysis of blood T cell subpopulations by flow cytometry. Blood cells were isolated from wild-type C57BL/6 and humanized B-hPD-1/hPD-L1/hC5AR1 mice (female, n=3, 6-week-old), and analyzed by flow cytometry to assess leukocyte subpopulations. (A) Representative flow cytometry plots. Single live CD45+ cells were gated on TCRβ+ T cells and used for further analysis as indicated. (B) Percent of CD4+ T cells, CD8+ T cells, and Tregs in B-hPD-1/hPD-L1/hC5AR1 mice were similar to those in wild-type mice, demonstrating that introduction of hPD-1, hPD-L1 and hC5AR1 in place of their murine counterparts does not change the overall development, differentiation or distribution of these blood T cell subtypes. Values are expressed as mean ± SEM.
-
Antibody binding analysis

-
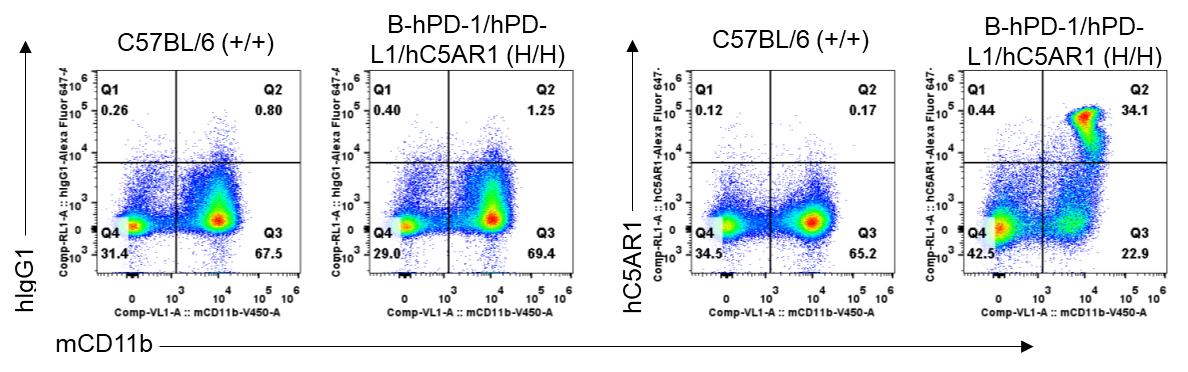
Antibody binding analysis of B-hPD-1/hPD-L1/hC5AR1 mice by flow cytometry. Splenocytes were isolated from wild-type C57BL/6 mice (+/+) and homozygous B-hPD-1/hPD-L1/hC5AR1 mice (H/H). Anti-hC5AR1 antibody avdoralimab (in house) can bind CD11b+ cells in the spleen of B-hPD-1/hPD-L1/hC5AR1 mice. In contrast, the antibody cannot bind cells isolated from the spleen of wild-type C57BL/6 mice. Thus the anti-hC5AR1 antibody can specifically bind cells from B-hPD-1/hPD-L1/hC5AR1 mice.
-
In vivo efficacy of combination of anti-human PD-1 antibody and anti-human C5AR1 antibody

-
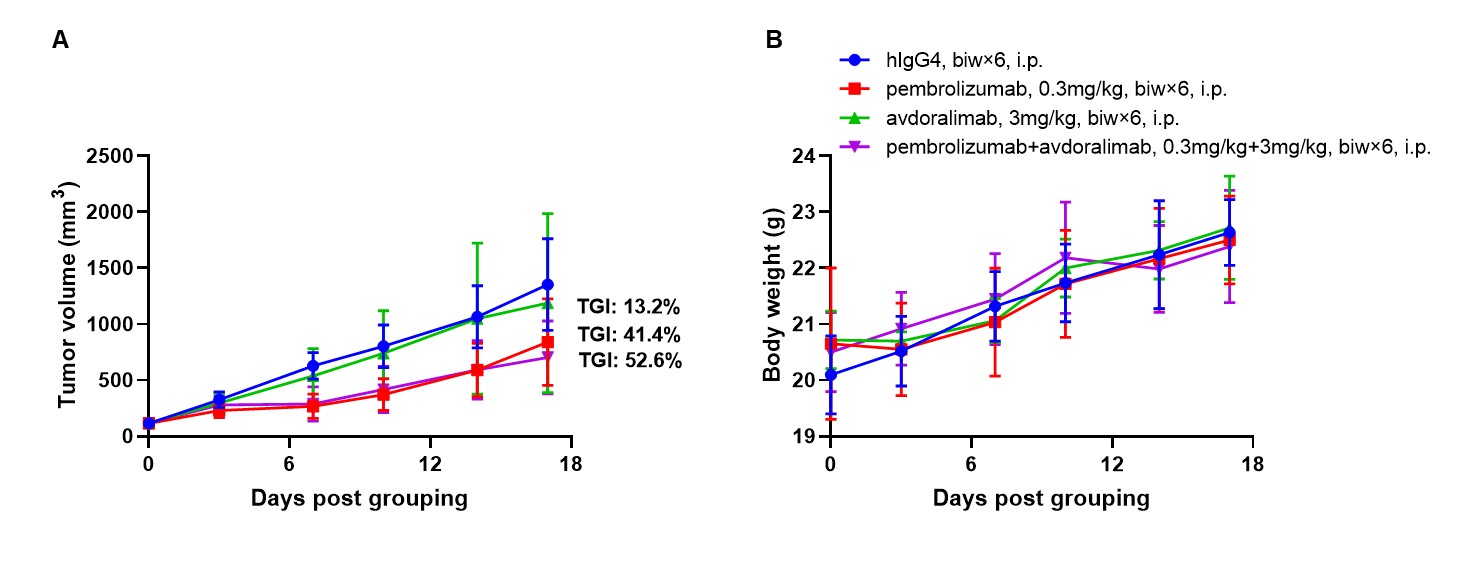
Antitumor activity of combination of anti-human PD-1 antibody (pembrolizumab, in house) and anti-human C5AR1 antibodies (avdoralimab, in house) in B-hPD-1/hPD-L1/hC5AR1 mice. (A) combination of anti-human PD-1 antibody and anti-human C5AR1 antibody inhibited B-hPD-L1 MC38 plus tumor growth in B-hPD-1/hPD-L1/hC5AR1 mice. Murine colon cancer B-hPD-L1 MC38 plus cells were subcutaneously implanted into homozygous B-hPD-1/hPD-L1/hC5AR1 mice (female, 7-9-week-old, n=6). Mice were grouped when tumor volume reached approximately 100 mm3, at which time they were treated with anti-human PD-1 antibody and anti-human C5AR1 antibody with doses and schedules indicated in panel. (B) Body weight changes during treatment. As shown in panel A, combination of anti-human PD-1 antibody and anti-human C5AR1 antibody were efficacious in controlling tumor growth in B-hPD-1/hPD-L1/hC5AR1 mice, demonstrating that the B-hPD-1/hPD-L1/hC5AR1 mice provide a powerful preclinical model for in vivo evaluation of combination of anti-human PD-1 antibody and anti-human C5AR1 antibody. Values are expressed as mean ± SEM.
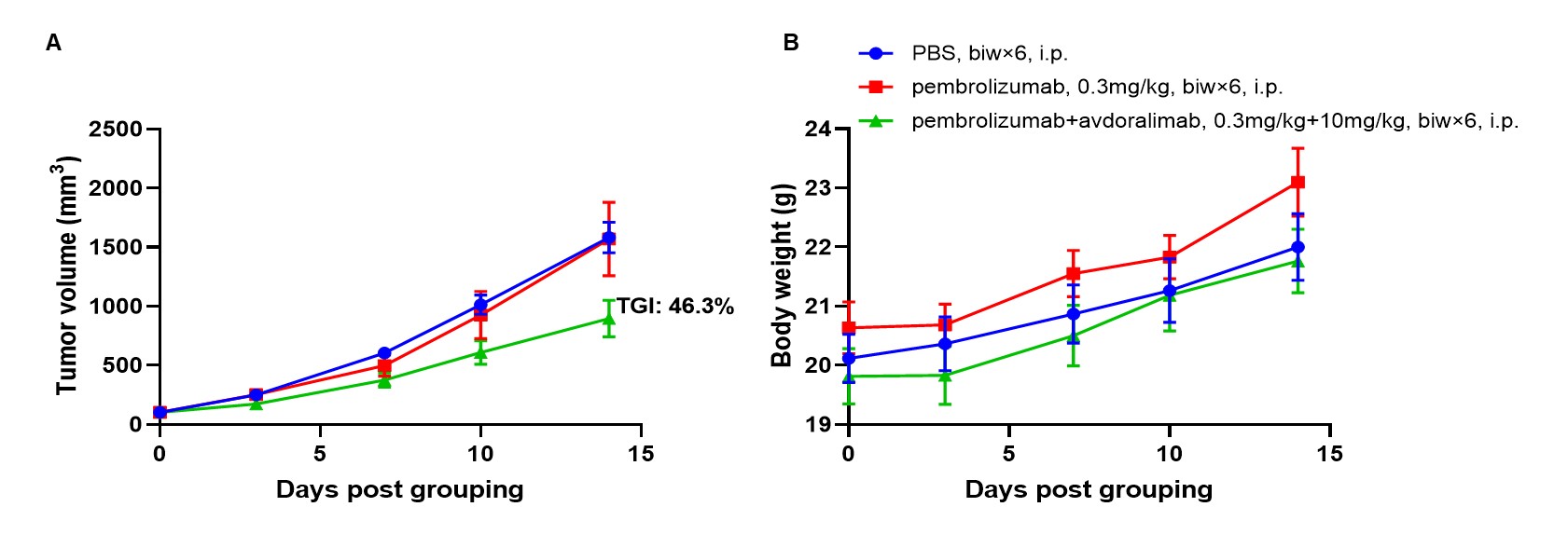
Antitumor activity of combination of anti-human PD-1 antibody (pembrolizumab, in house) and anti-human C5AR1 antibodies (avdoralimab, in house) in B-hPD-1/hPD-L1/hC5AR1 mice. (A) combination of anti-human PD-1 antibody and anti-human C5AR1 antibody inhibited B-hPD-L1 MC38 plus tumor growth in B-hPD-1/hPD-L1/hC5AR1 mice. Murine colon cancer B-hPD-L1 MC38 plus cells were subcutaneously implanted into homozygous B-hPD-1/hPD-L1/hC5AR1 mice (female, 5-8-week-old, n=6). Mice were grouped when tumor volume reached approximately 100 mm3, at which time they were treated with anti-human PD-1 antibody and anti-human C5AR1 antibody with doses and schedules indicated in panel. (B) Body weight changes during treatment. As shown in panel A, combination of anti-human PD-1 antibody and anti-human C5AR1 antibody were efficacious in controlling tumor growth in B-hPD-1/hPD-L1/hC5AR1 mice, demonstrating that the B-hPD-1/hPD-L1/hC5AR1 mice provide a powerful preclinical model for in vivo evaluation of combination of anti-human PD-1 antibody and anti-human C5AR1 antibody. Values are expressed as mean ± SEM.


