Basic Information
-
Details

-
Protein expression in splenocytes
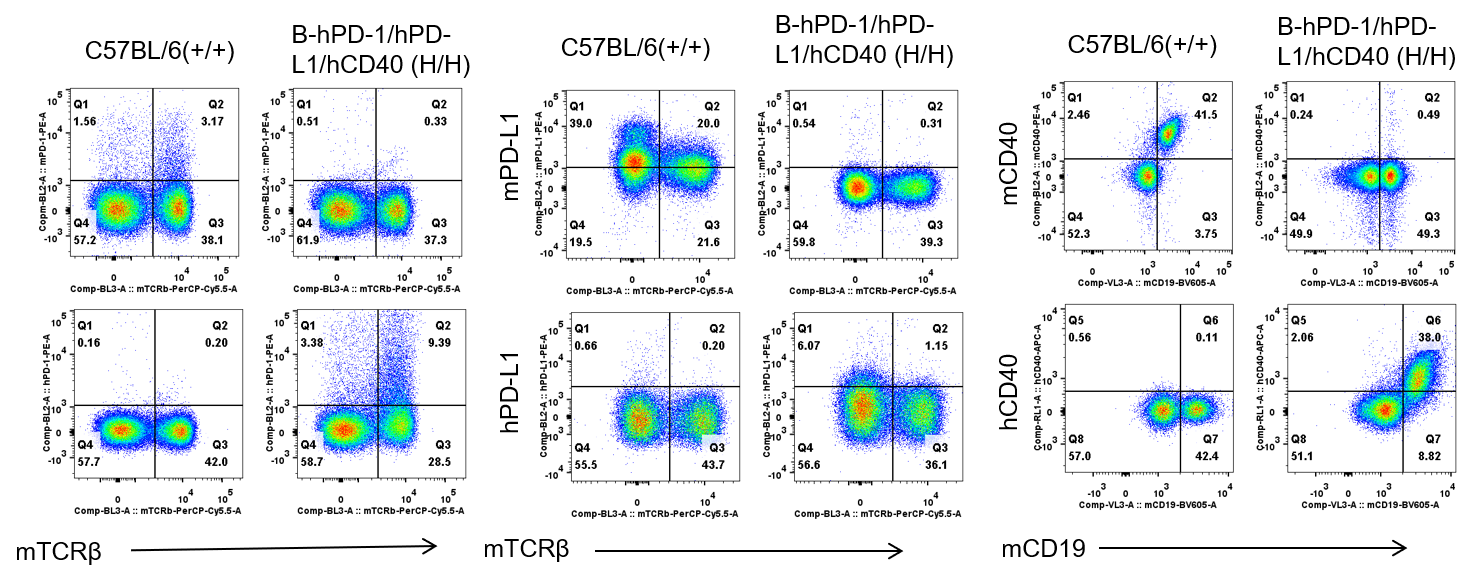
Strain specific PD-1, PD-L1 and CD40 expression analysis in homozygous B-hPD-1/hPD-L1/hCD40 mice by flow cytometry. Splenocytes were collected from WT and homozygous B-hPD-1/hPD-L1/hCD40 (H/H) mice analyzed by flow cytometry with species-specific anti-PD-1 antibody. Mouse PD-1, PD-L1 and CD40 were detectable in WT mice. Human PD-1, PD-L1 and CD40 were exclusively detectable in homozygous B-hPD-1/hPD-L1/hCD40 but not WT mice.
Protein expression analysis in stimulated splenocytes
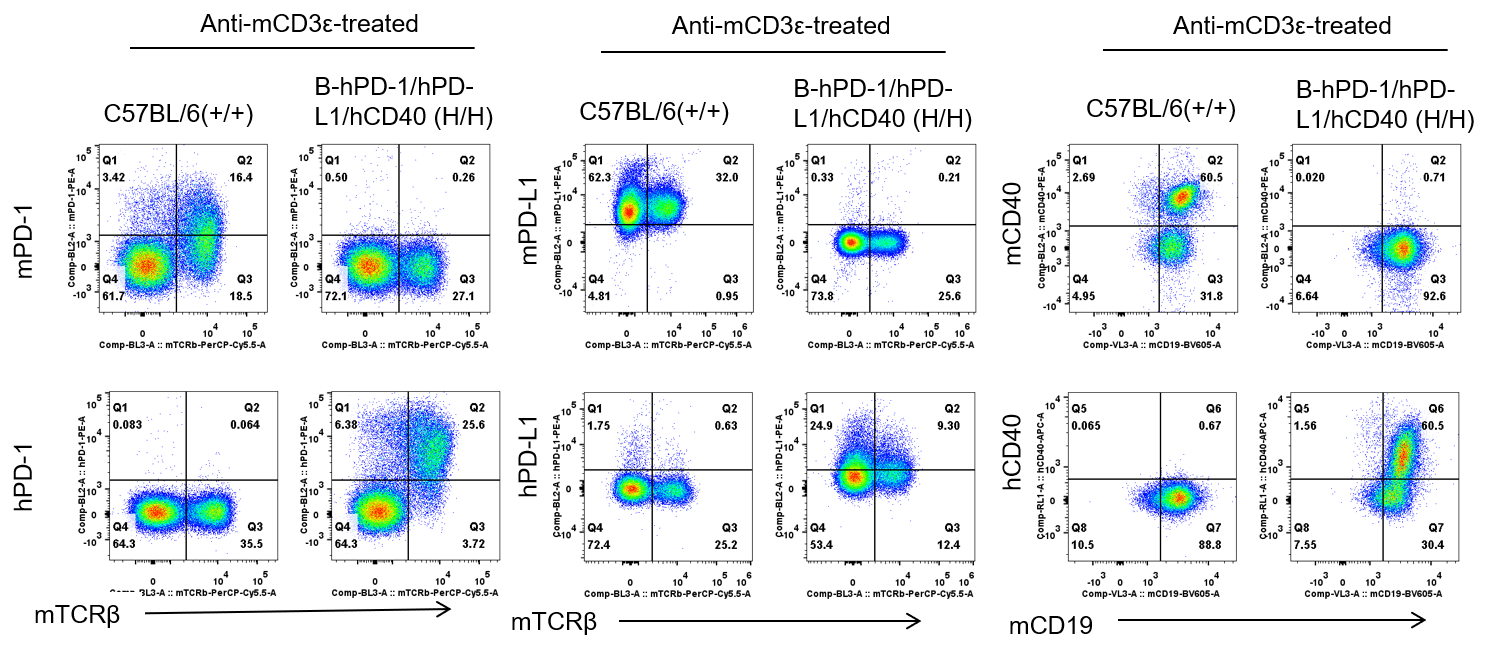
Strain specific PD-1, PD-L1 and CD40 expression analysis in homozygous B-hPD-1/hPD-L1/hCD40 mice by flow cytometry. Splenocytes were collected from WT and homozygous B-hPD-1/hPD-L1/hCD40 (H/H) mice after stimulation with anti-CD3ε in vivo (7.5 μg/mice), and without stimulation, and analyzed by flow cytometry with species-specific anti-PD-1 antibody. Mouse PD-1, PD-L1 and CD40 were detectable in WT mice. Human PD-1, PD-L1 and CD40 were exclusively detectable in homozygous B-hPD-1/hPD-L1/hCD40 but not WT mice. After anti-CD3ε stimulation, hPD-1 protein expression was significantly increased.
Combination therapy of Pembrolizumab and Selicrelumab
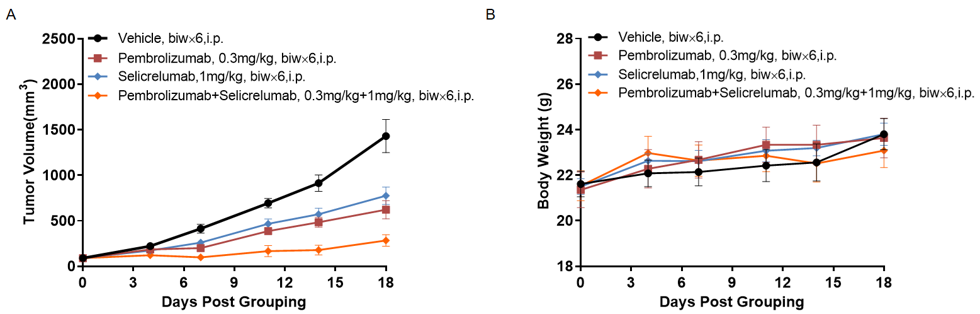
Antitumor activity of of Pembrolizumab and Selicrelumab in B-hPD-1/hPD-L1/hCD40 mice. (A) anti-human PD-1 antibody combined with anti-human CD40 antibody inhibited MC38-hPD-L1 tumor growth in B-hPD-1/hPD-L1/hCD40 mice. Murine colon cancer MC38-hPD-L1 cells (5×105) were subcutaneously implanted into homozygous B-hPD-1/hPD-L1/hCD40 mice (female, 7 week-old, n=5). Mice were grouped when tumor volume reached approximately 100-150 mm3, at which time they were treated with Pembrolizumab and Selicrelumab with doses and schedules indicated in panel A. (B) Body weight changes during treatment. As shown in panel A, combination of Pembrolizumab and Selicrelumab shows more inhibitory effects than individual groups, demonstrating that the B-hPD-1/hPD-L1/hCD40 mice provide a powerful preclinical model for in vivo evaluating combination therapy efficacy of PD-1 antibodies combined with anti-human CD40 antibodies. Values are expressed as mean ± SEM.(All antibodies were made in-house)


