Basic Information
-
Targeting strategy

-
Gene targeting strategy for B-hPD-1/hPD-L1/hIL2RA mice. The exon 2 of mouse Pd-1 gene that encodes the extracellular domain was replaced by human PD-1 exon 2 in B-hPD-1/hPD-L1/hIL2RA mice. The exon 3 of mouse Pd-l1 gene that encodes the extracellular domain was replaced by human PD-L1 exon 3 in B-hPD-1/hPD-L1/hIL2RA mice. The exons 2-6 of mouse Il2ra gene that encode the extracellular domain were replaced by human IL2RA exons 2-6 in B-hPD-1/hPD-L1/hIL2RA mice.
-
Protein expression analysis in T cells

-
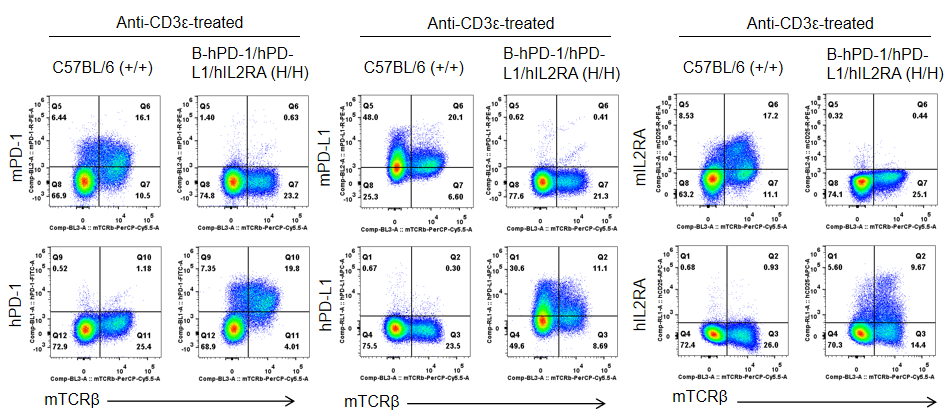
Strain specific PD-1, PDL1 and IL2RA expression analysis in homozygous B-hPD-1/hPD-L1/hIL2RA mice by flow cytometry. Splenocytes were collected from WT and homozygous B-hPD-1/hPD-L1/hIL2RA mice stimulated with anti-CD3ε in vivo, and analyzed by flow cytometry with species-specific PD-1, PD-L1 and IL2RA antibody. Mouse PD-1, PD-L1 and IL2RA were detectable in WT mice. Human PD-1, PD-L1 and IL2RA were exclusively detectable in homozygous B-hPD-1/hPD-L1/hIL2RA but not WT mice.
-
Protein expression analysis of IL2RA in Tregs

-
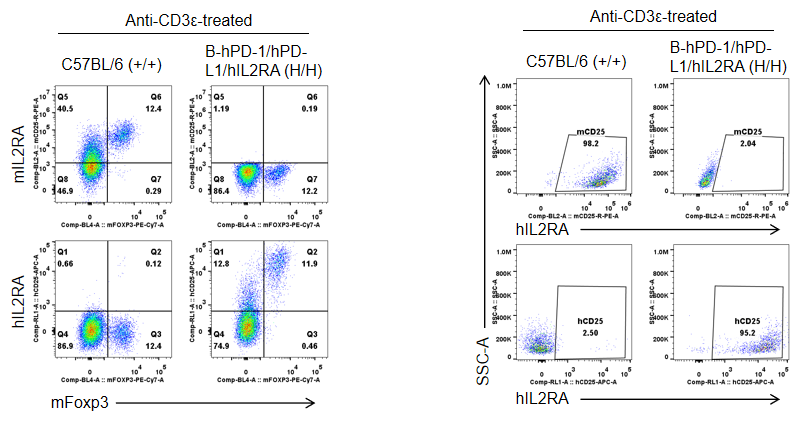
Strain specific IL2RA expression analysis in homozygous B-hPD-1/hPD-L1/hIL2RA mice by flow cytometry. Splenocytes were collected from WT and homozygous B-hPD-1/hPD-L1/hIL2RA mice stimulated with anti-CD3ε in vivo, and analyzed by flow cytometry with species-specific IL2RA antibody. Mouse IL2RA were detectable in WT mice. Human IL2RA were exclusively detectable in homozygous B-hPD-1/hPD-L1/hIL2RA but not WT mice.


