Basic Information
-
Targeting Strategy

-
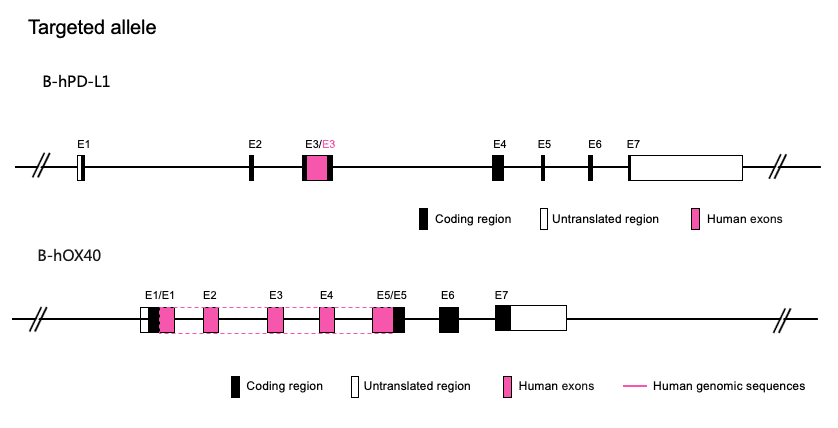
Gene targeting strategy for B-hPD-L1/hOX40 mice. The exon 3 of mouse Pd-l1 gene that encodes the extracellular domain was replaced by human PD-L1 exon 3 and the exons 1-5 of mouse Tnfrsf4 gene that encode the extracellular domain were replaced by human TNFRSF4 exons 1-5 in B-hPD-L1/hOX40 mice.
-
Protein expression analysis

-
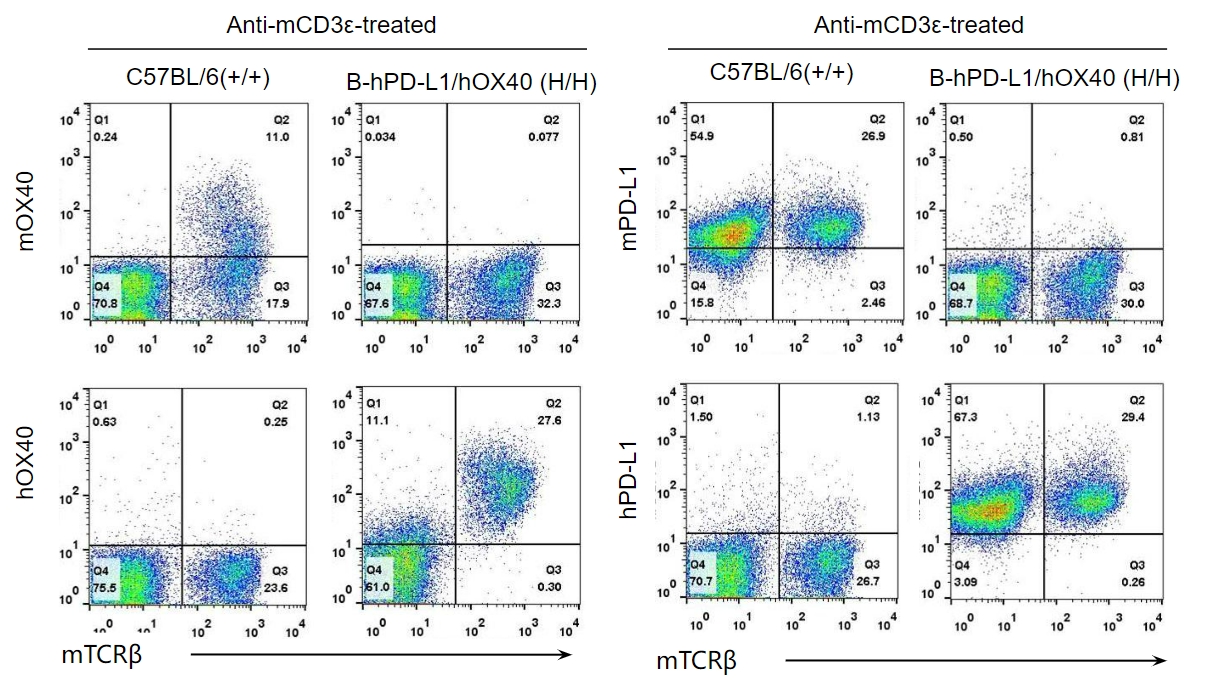
Strain specific OX40 and PD-L1 expression analysis in homozygous B-hPD-L1/hOX40 mice by flow cytometry. Splenocytes were collected from WT and homozygous B-hPD-L1/hOX40 (H/H) mice stimulated with anti-CD3ε in vivo (7.5 μg/mice), and analyzed by flow cytometry with species-specific anti-OX40 antibody. Mouse OX40 and PD-L1 were exclusively detected in WT mice. Human OX40 and PD-L1 were exclusively detected in homozygous B-hPD-L1/hOX40 but not WT mice.
-
Combination therapy of PD-L1 Ab and OX40 Ab

-
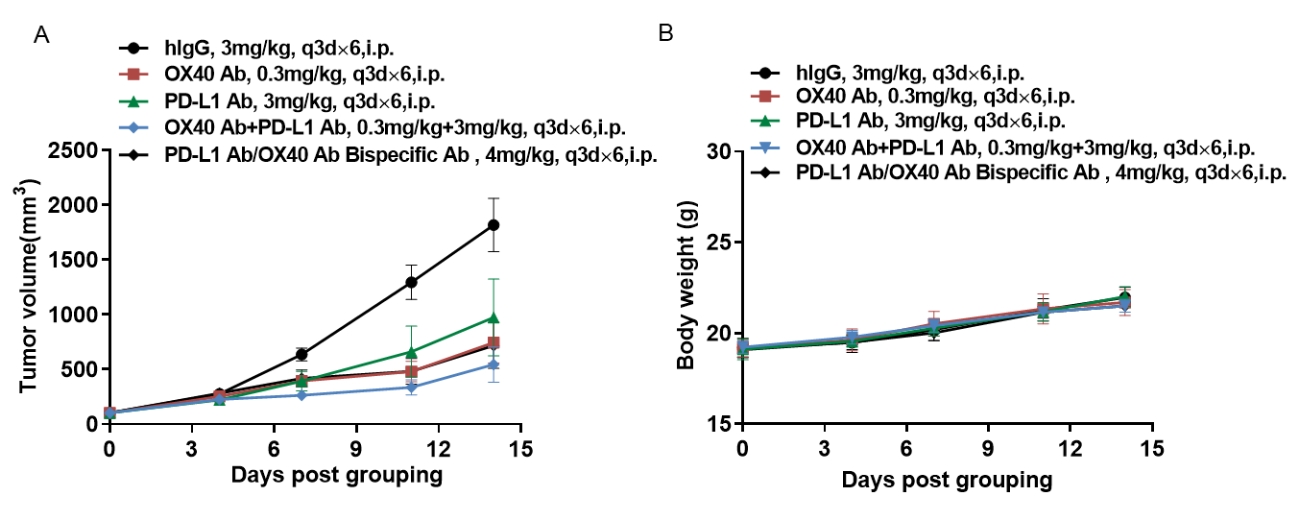
Antitumor activity of anti-hOX40 antibody combined with anti-hPD-L1 antibody in B-hPD-L1/hOX40 mice. (A) Anti-hOX40 antibody combined with anti-hPD-L1 antibodies inhibited MC38-hPD-L1 tumor growth in B-hPD-L1/hOX40 mice. Murine colon cancer MC38-hPD-L1 cells (5×105) were subcutaneously implanted into homozygous B-hPD-L1/hOX40 mice (female, 6-7 week-old, n=6). Mice were grouped when tumor volume reached approximately 150±50 mm3, at which time they were treated with anti-hOX40 antibody combined with anti-hPD-L1 antibody with doses and schedules indicated in panel (B) Body weight changes during treatment. As shown in panel A, combination of anti-hOX40 and anti-hPD-L1 antibody shows more inhibitory effects than individual groups, demonstrating that the B-hPD-L1/hOX40 mice provide a powerful preclinical model for in vivo evaluating combination therapy efficacy of hOX40 antibodies and hPD-L1 antibodies. Values are expressed as mean ± SEM.


