Basic Information
-
Gene targeting strategy

-
Gene targeting strategy for B-hPSGL-1 mice. The exon 2 of mouse Selplg gene that encodes the extracellular domain was replaced by human SELPLG exon 2 in B-hPSGL-1 mice.
-
Protein expression analysis

-
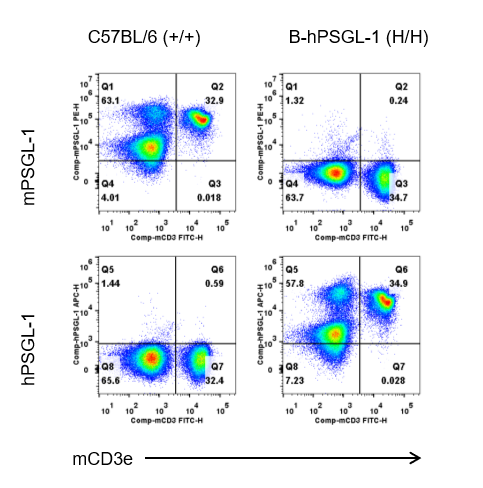
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H), and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable in wild-type C57BL/6 mice (+/+). Human PSGL-1 was exclusively detectable in homozygous B-hPSGL-1 mice (H/H) but not wild-type C57BL/6 mice (+/+).
-
Protein expression analysis-spleen T cells

-
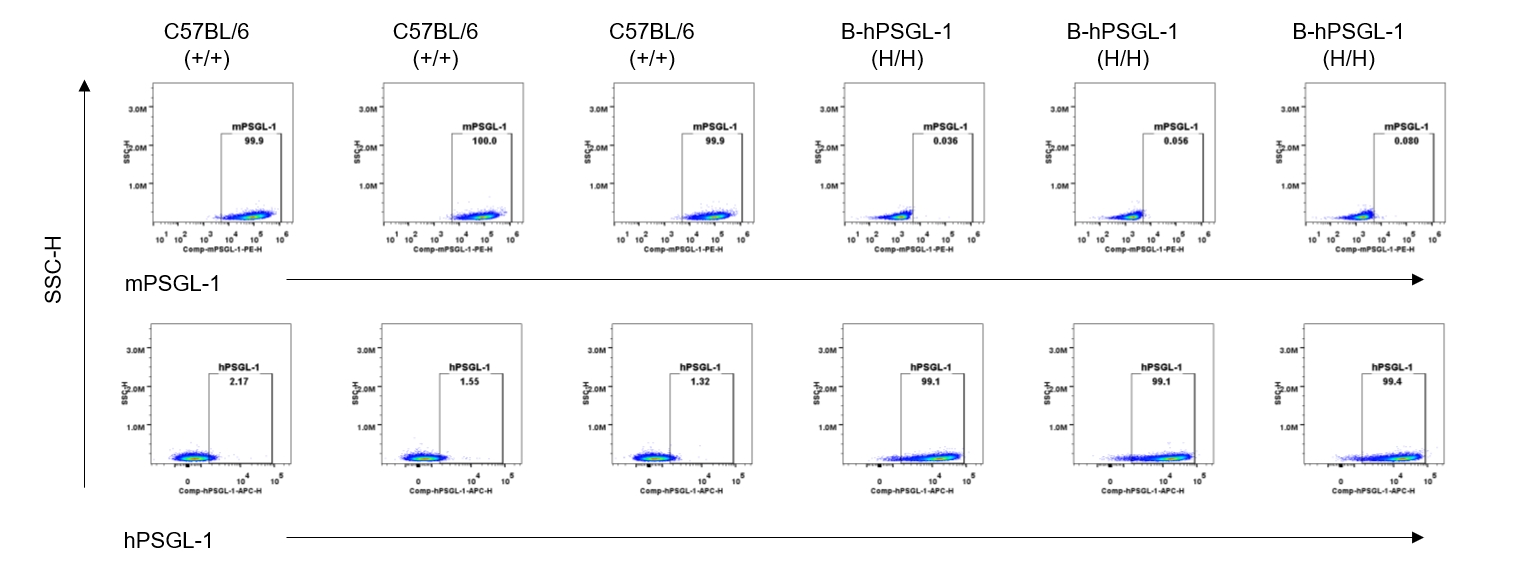
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on T cells of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on T cells of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-spleen B cells

-
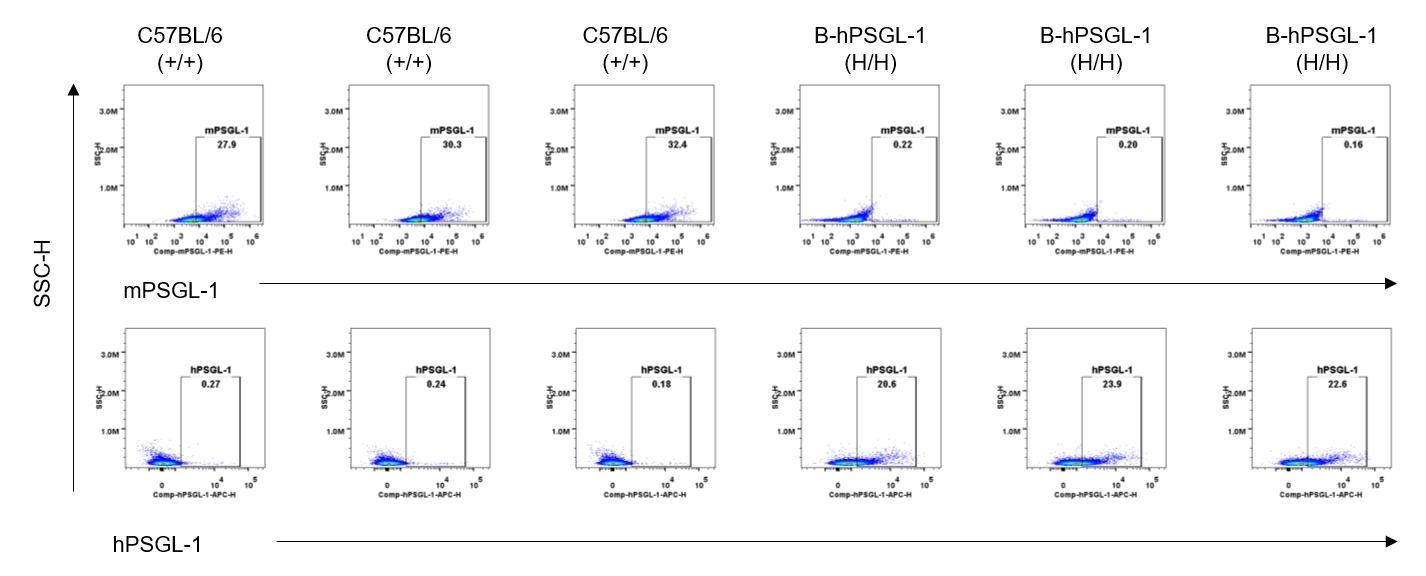
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry.Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on B cells of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on B cells of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-spleen NK cells

-
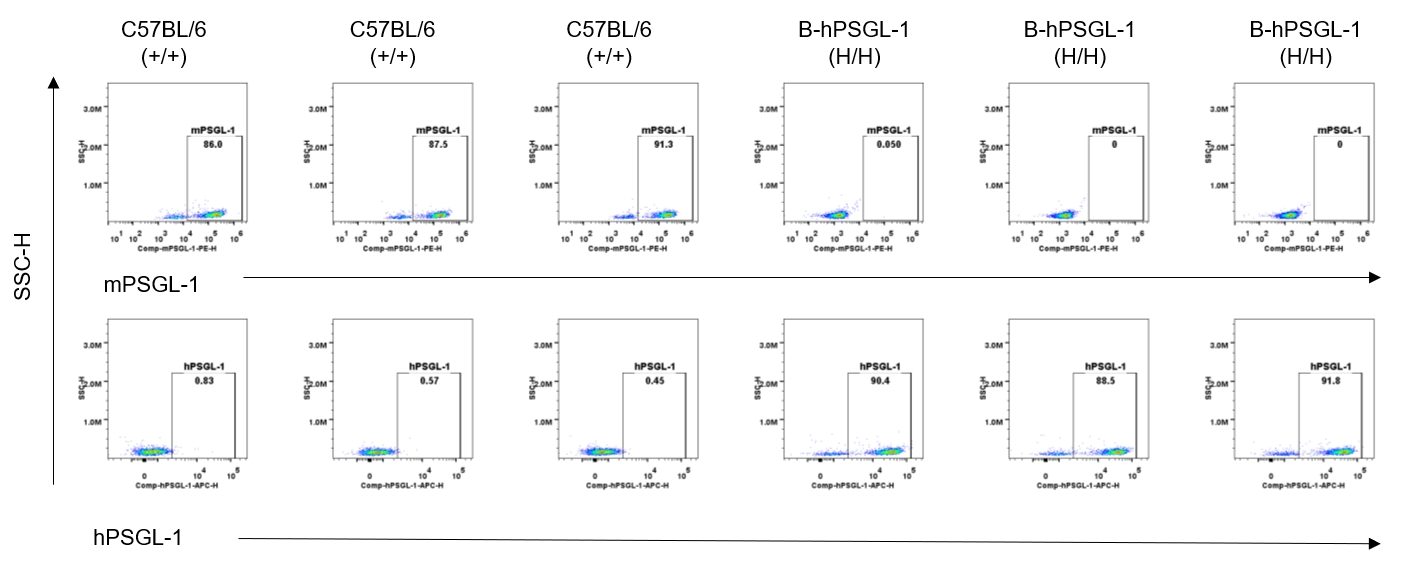
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on NK cells of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on NK cells of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-spleen DCs

-
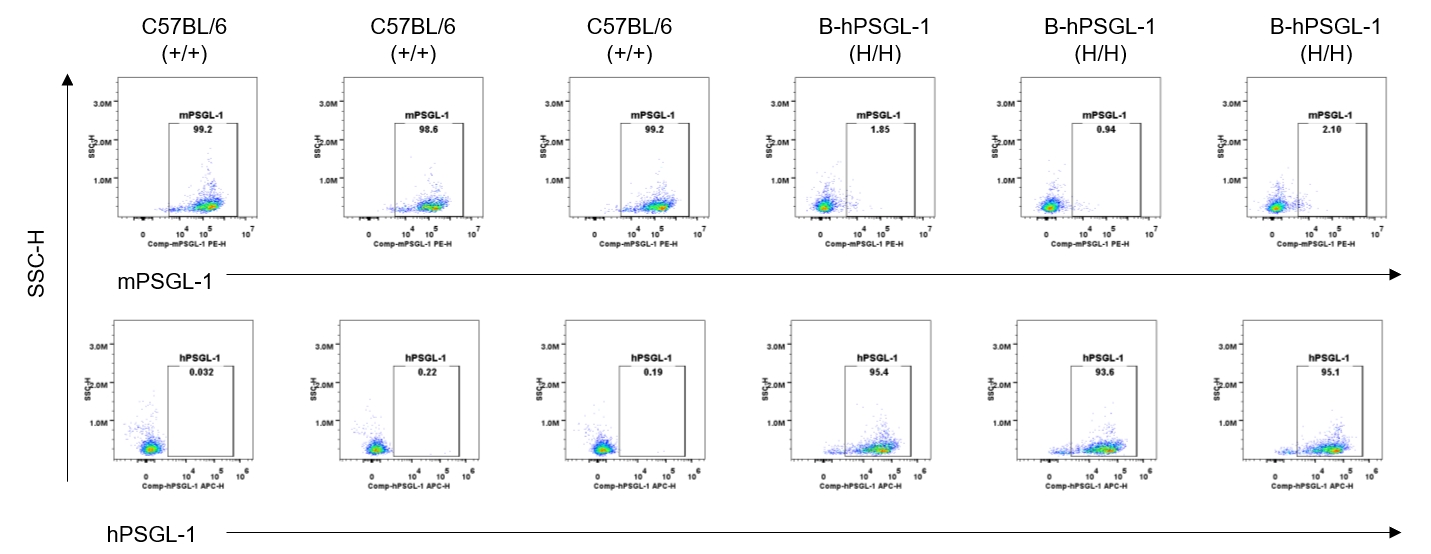
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on dendritic cells of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on dendritic cells of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-spleen neutrophils

-
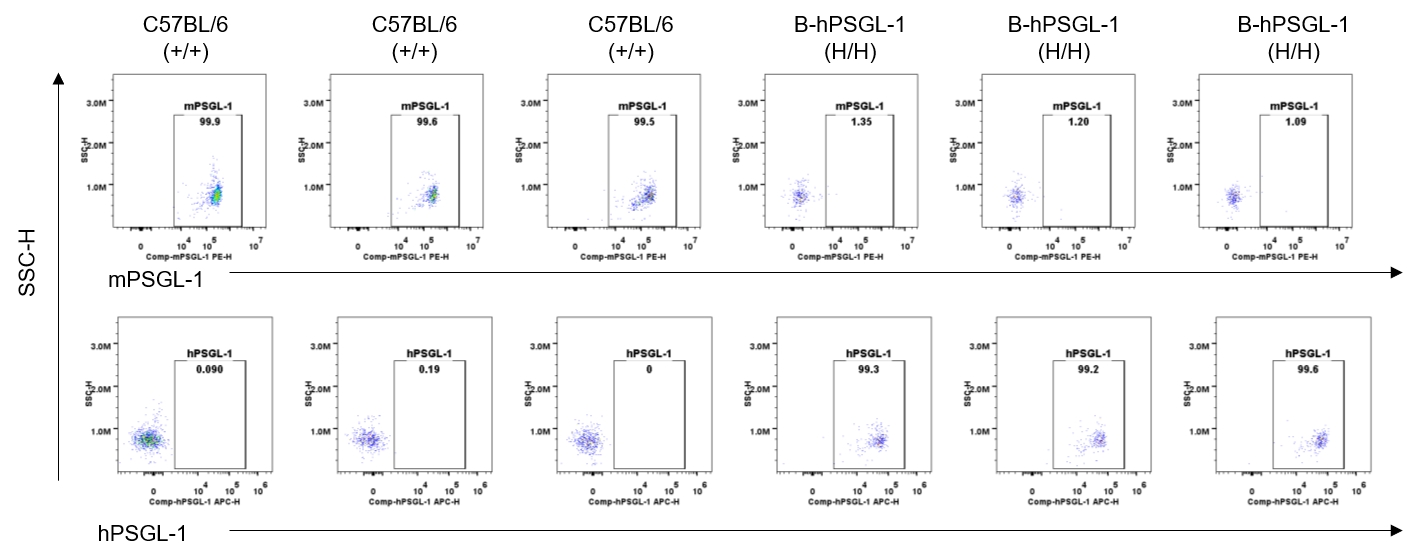
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on neutrophils of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on neutrophils of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-spleen monocytes

-
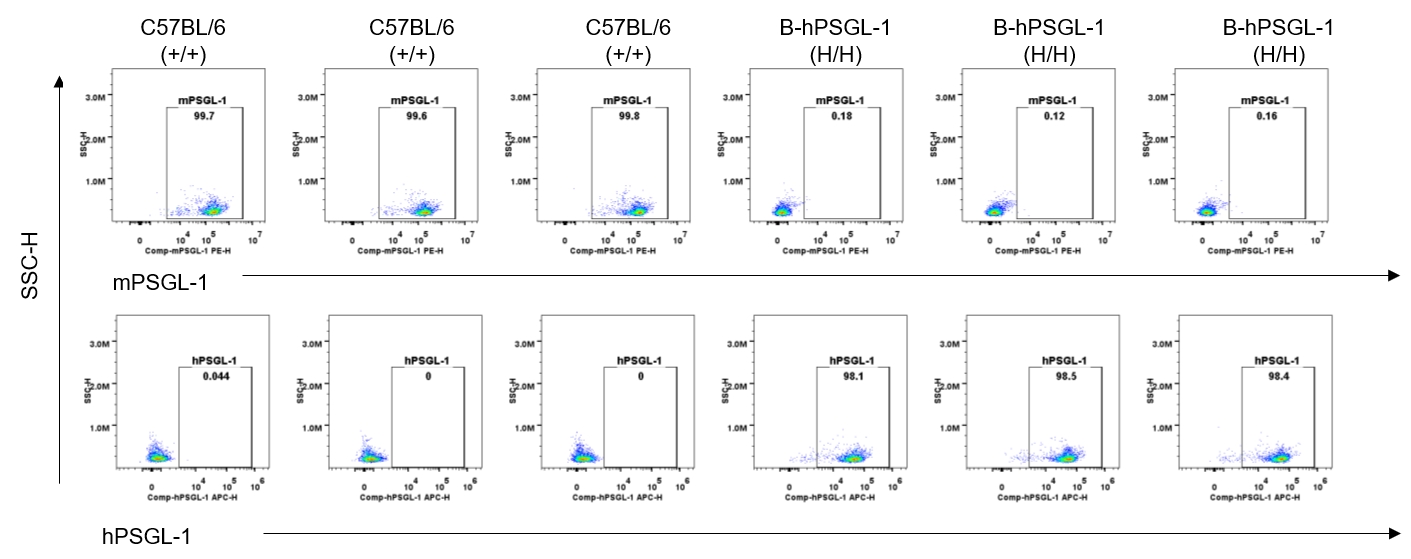
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on monocytes of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on monocytes of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-spleen macrophages

-
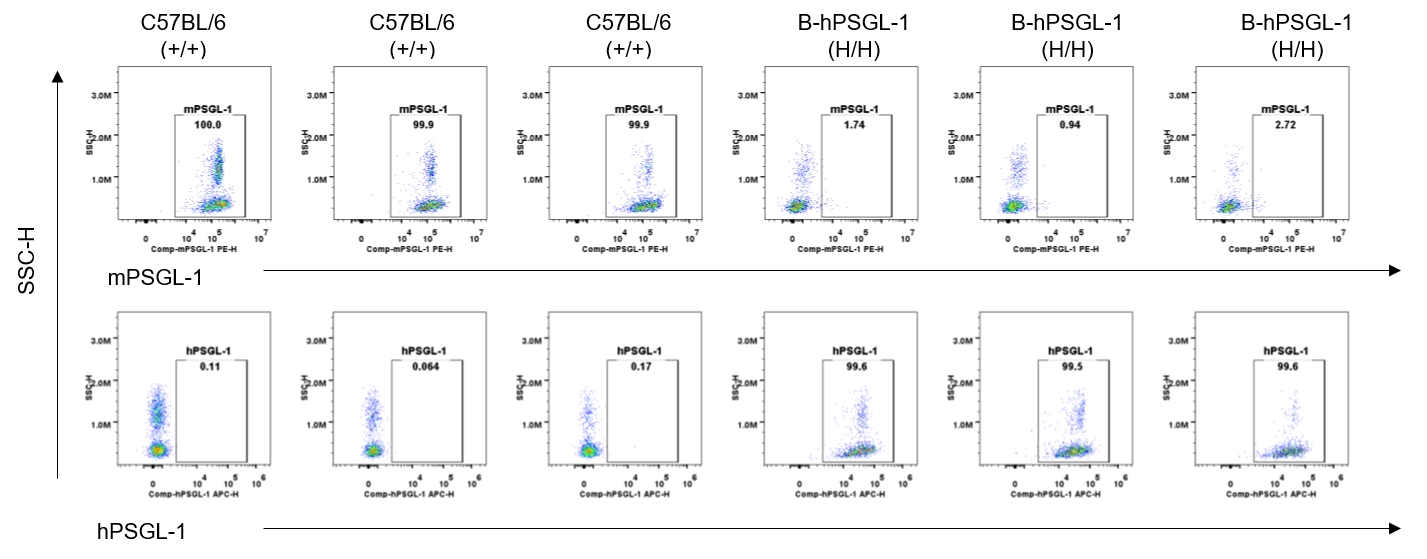
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on macrophages of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on macrophages of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-blood T cells

-
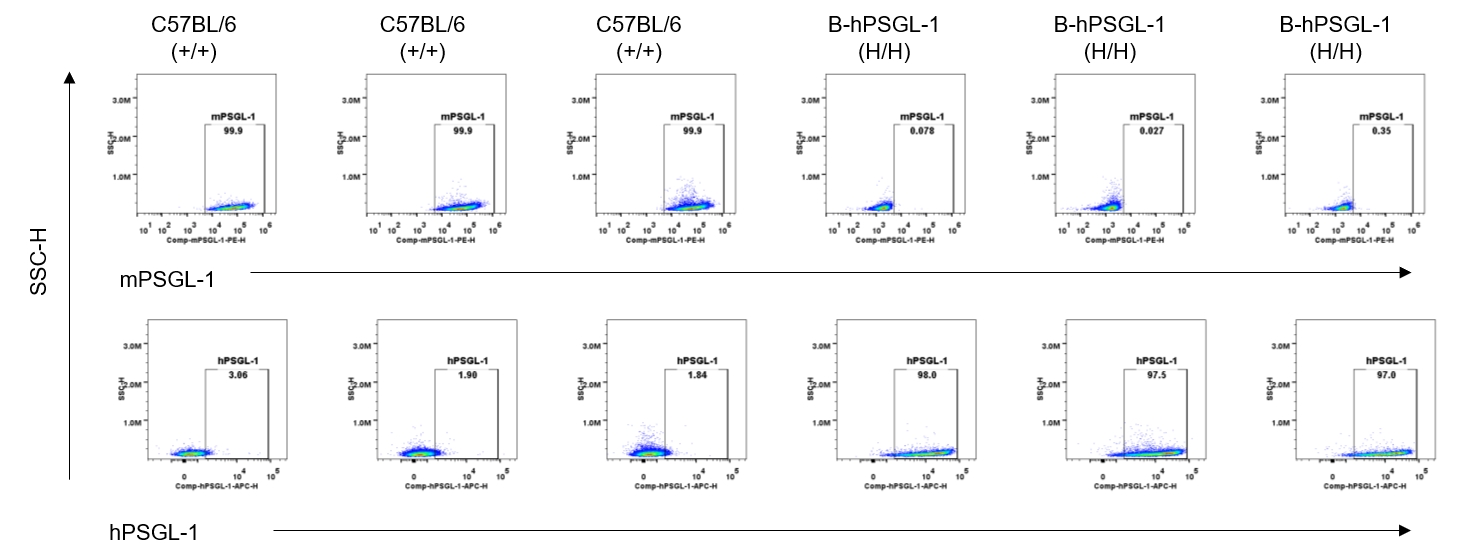
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Blood were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on T cells of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on T cells of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-blood B cells

-
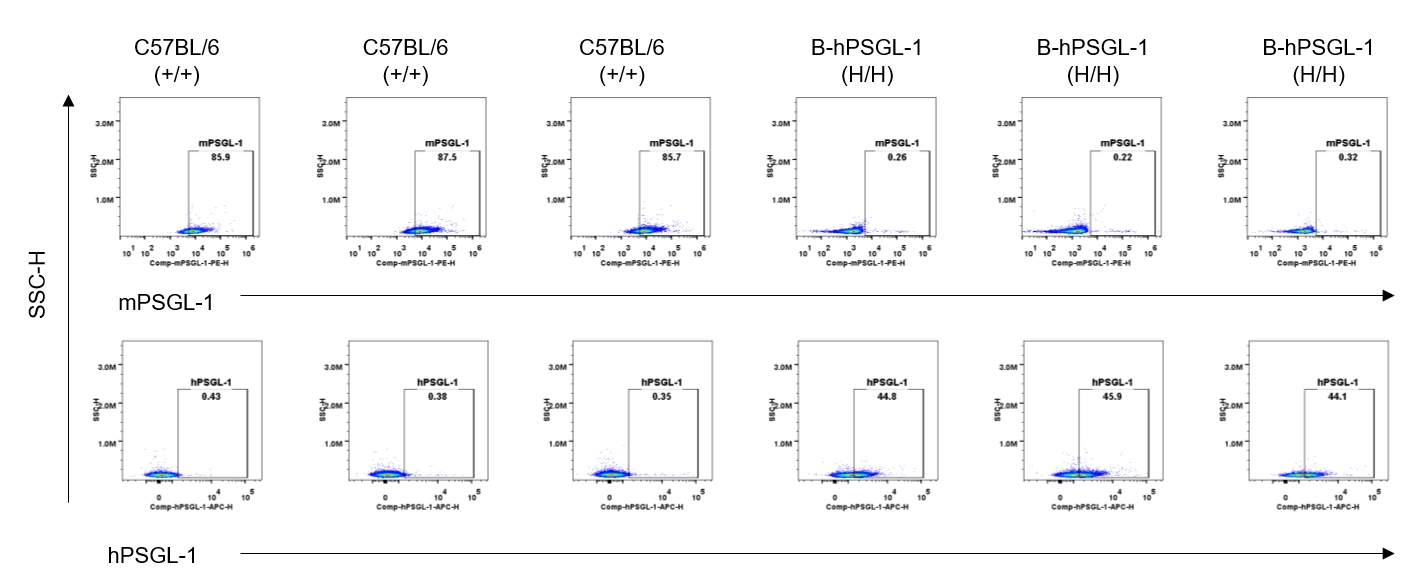
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Blood were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on B cells of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on B cells of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-blood NK cells

-
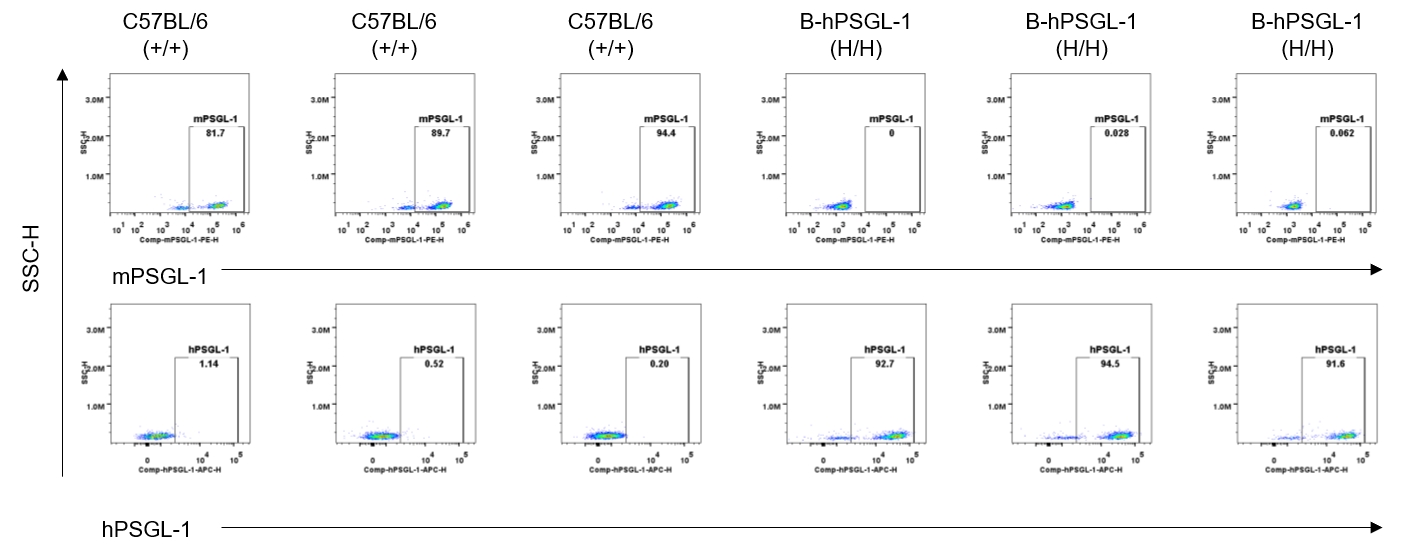
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Blood were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on NK cells of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on NK cells of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-blood DCs

-
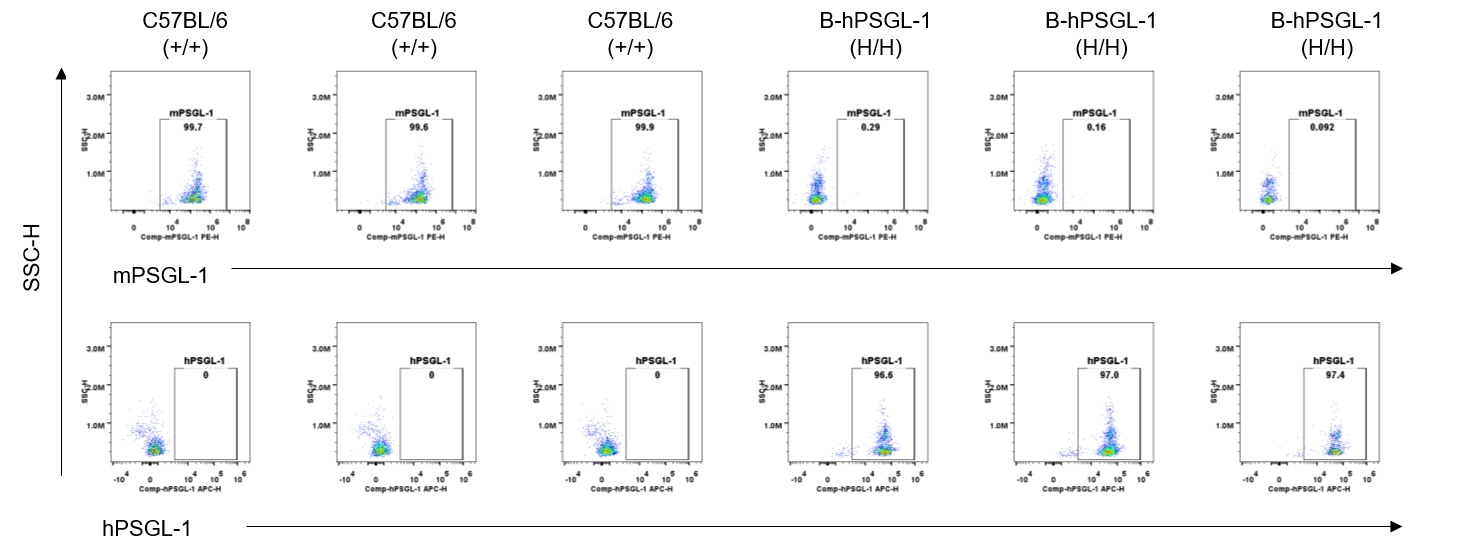
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Blood were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on dendritic cells of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on dendritic cells of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-blood neutrophils

-
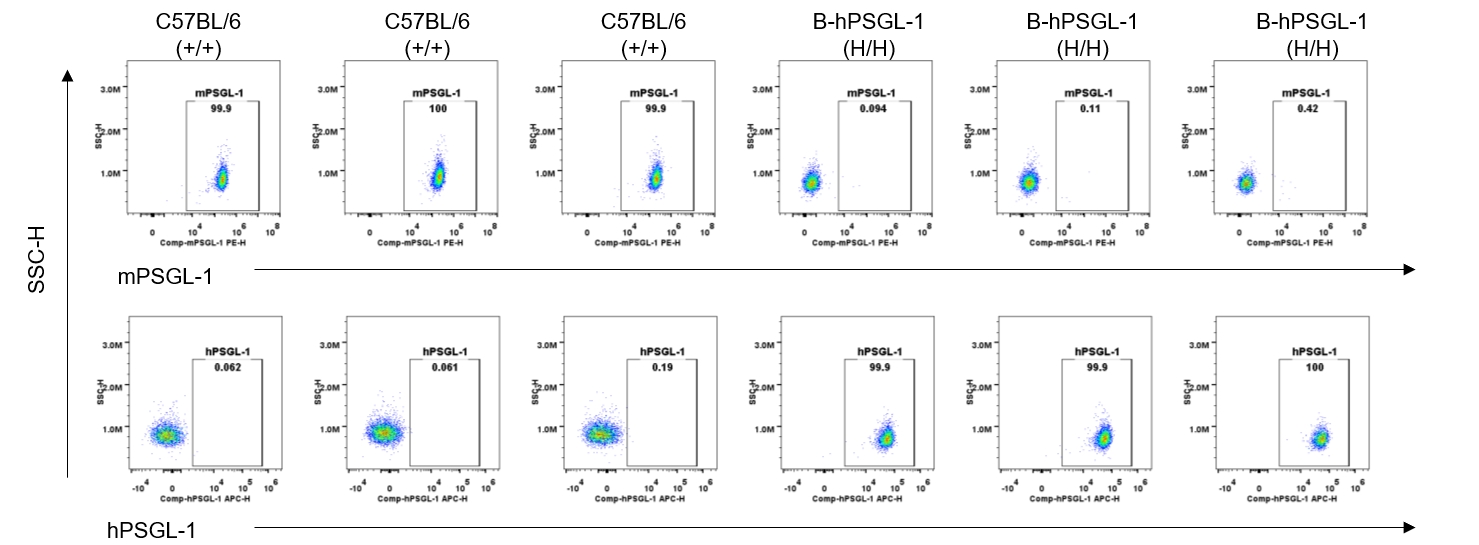
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Blood were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on neutrophils of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on neutrophils of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-blood monocytes

-
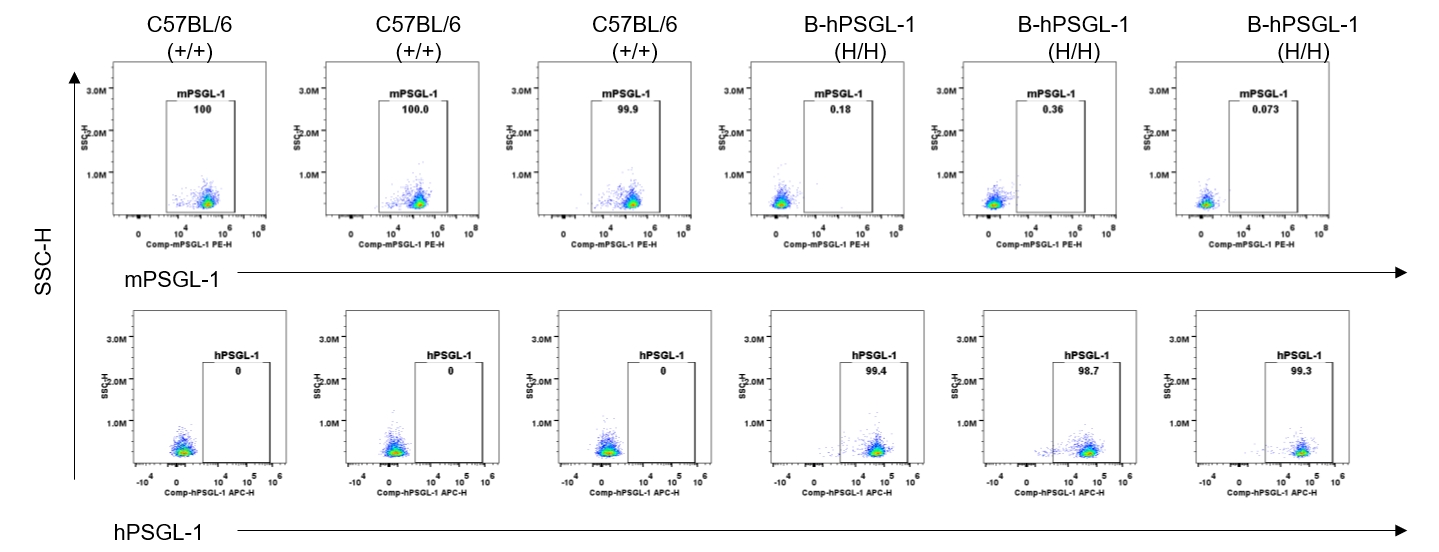
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Blood were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on monocytes of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on monocytes of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Protein expression analysis-blood macrophages

-
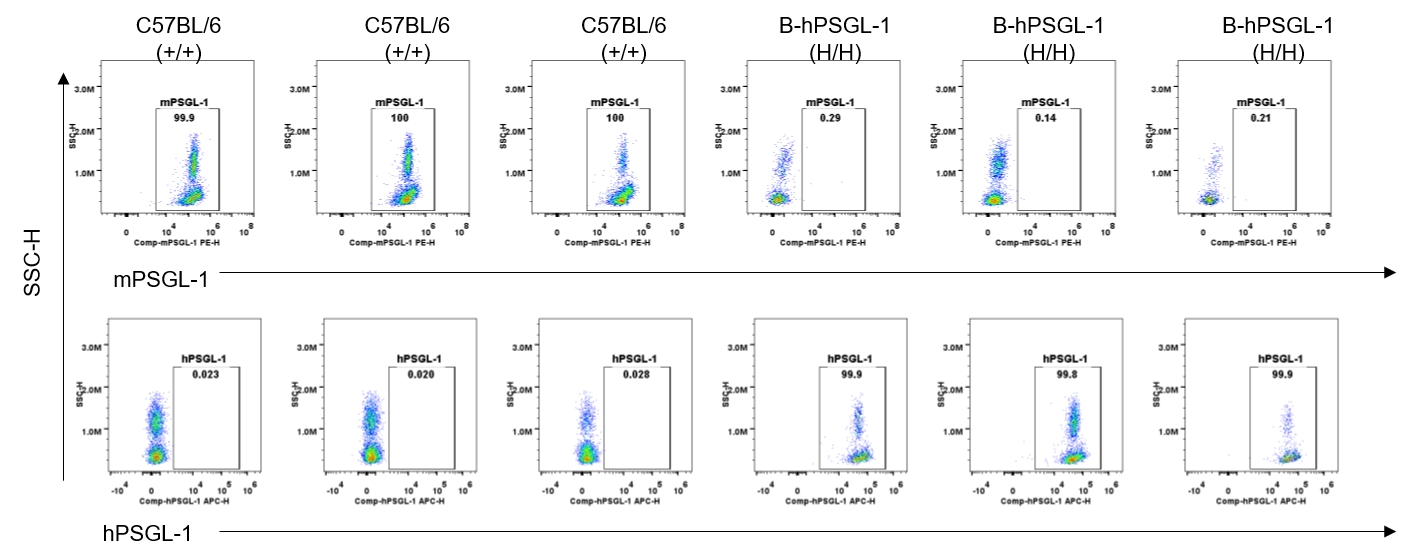
Strain specific PSGL-1 expression analysis in homozygous B-hPSGL-1 mice by flow cytometry. Blood were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hPSGL-1 mice (H/H) and analyzed by flow cytometry with species-specific anti-PSGL-1 antibody. Mouse PSGL-1 was detectable on macrophages of wild-type C57BL/6 mice. Human PSGL-1 was detectable only on macrophages of homozygous B-hPSGL-1 mice but not on wild-type mice.
-
Frequency of leukocyte subpopulations in spleen

-
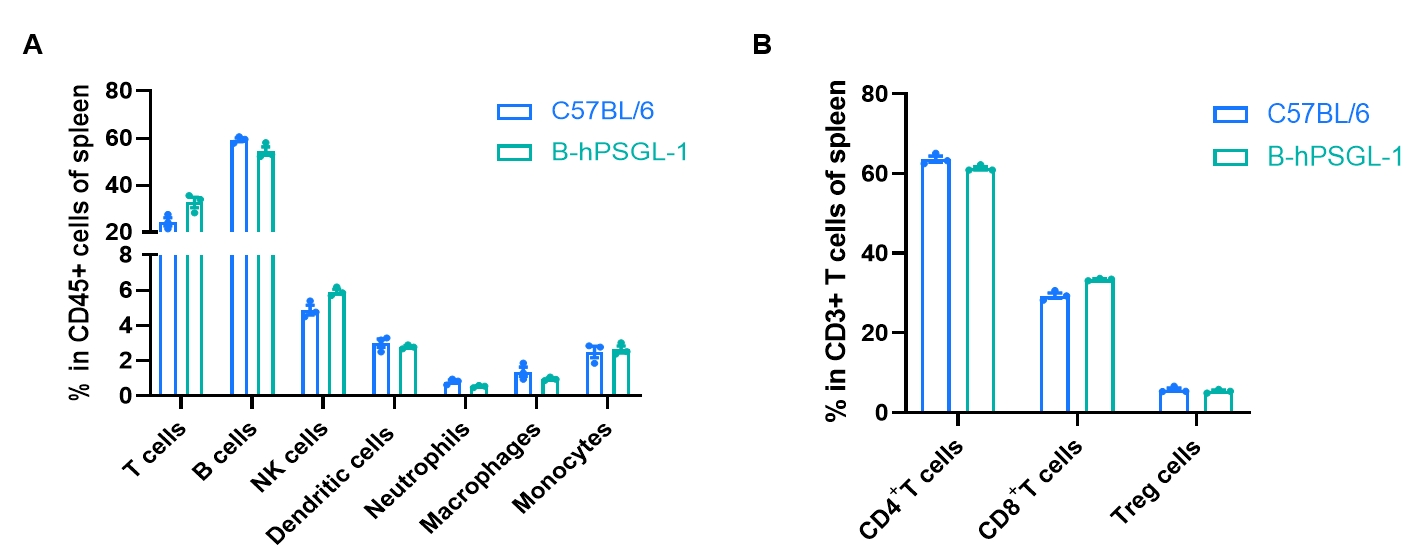
Frequency of leukocyte subpopulations in spleen by flow cytometry. Splenocytes were isolated from wild-type C57BL/6 mice and homozygous B-hPSGL-1 mice (n=3, 7-week-old). A. Flow cytometry analysis of the splenocytes was performed to assess the frequency of leukocyte subpopulations. B. Frequency of T cell subpopulations. Percentages of T cells, B cells, NK cells, dendritic cells, neutrophils, monocytes, macrophages, CD4+ T cells, CD8+ T cells and Tregs in B-hPSGL-1 mice were similar to those in C57BL/6 mice, demonstrating that humanization of PSGL-1 does not change the frequency or distribution of these cell types in spleen. The frequency of leukocyte subpopulations in lymph nodes and blood of B-hPSGL-1 mice were also comparable to wild-type C57BL/6 mice (Data not shown). Values are expressed as mean ± SEM. Significance was determined by two-way ANOVA test. *P < 0.05, **P < 0.01, ***p < 0.001.
-
Summary

-
Protein expression analysis: Human PSGL-1 was exclusively detectable of homozygous B-hPSGL-1 mice (H/H) but not in WT mice (+/+), and mouse PSGL-1 was detectable only in WT mice (+/+).


