Basic Information
-
Targeting strategy

-
Gene targeting strategy for B-hCCR8 mice. The exon 2 that encoding the full coding sequence of mouse Ccr8 gene was replaced by exon 2 of human CCR8 gene in B-hCCR8 mice.
-
mRNA expression analysis

-
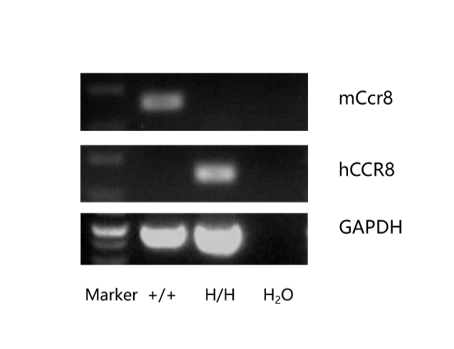
Strain specific analysis of CCR8 gene expression in C57BL/6 and B-hCCR8 mice by RT-PCR. Mouse Ccr8 mRNA was detectable in thymocytes of wild-type mice (+/+) . Human CCR8 mRNA was detectable only in homozygous B-hCCR8 mice (H/H) but not in wild-type mice (+/+).
-
Protein expression analysis of mouse and human CCR8 in spleen

-
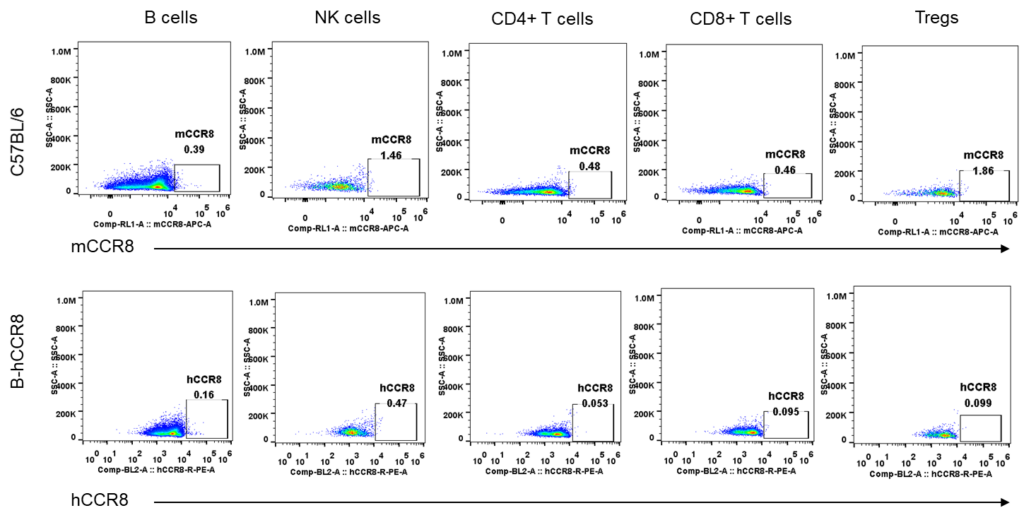
Strain specific analysis of CCR8 gene expression in B-hCCR8 mice by FACS. Spleen was isolated from the mice, and analyzed by flow cytometry. Human and mouse CCR8 were not expressed neither in splenocytes of wild-type mice nor B-hCCR8 mice separately.
-
Protein expression analysis of mouse and human CCR8 in thymus

-
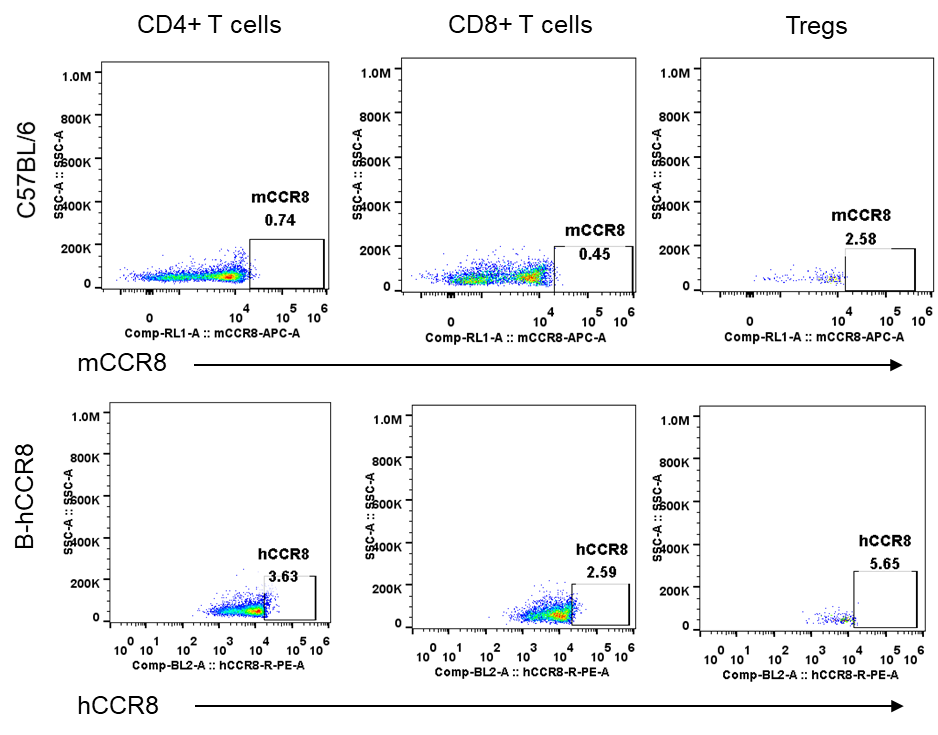
Strain specific analysis of CCR8 gene expression in B-hCCR8 mice by FACS. Thymus was isolated from the mice, and analyzed by flow cytometry. Human and mouse CCR8 were not expressed neither in thymocytes of wild-type mice nor B-hCCR8 mice separately.
-
Protein expression analysis of mouse and human CCR8 in blood

-
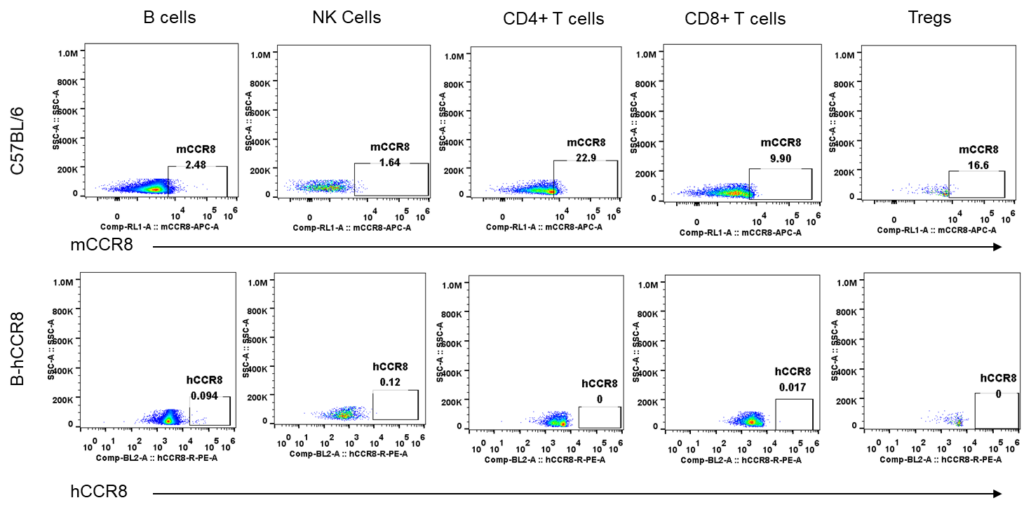
Strain specific analysis of CCR8 gene expression in B-hCCR8 mice by FACS. Blood were harvested from the mice, and analyzed by flow cytometry. Human and mouse CCR8 were not expressed neither in blood cells of wild-type mice nor B-hCCR8 mice separately.
-
Analysis of leukocytes cell subpopulation in B-hCCR8 mice

-
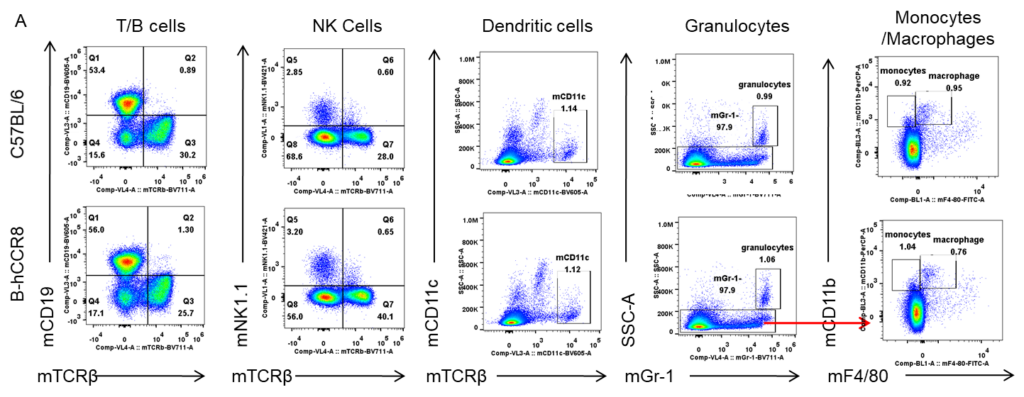
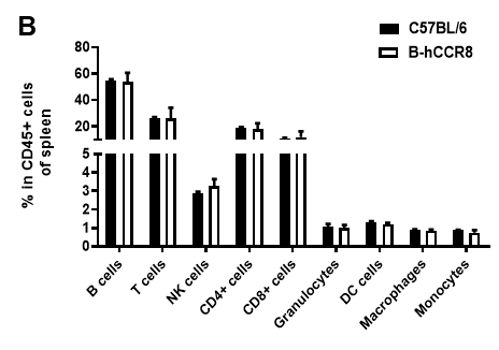
Analysis of spleen leukocyte subpopulations by FACS. Splenocytes were isolated from female C57BL/6 and B-hCCR8 mice (n=3, 8-week-old). Flow cytometry analysis of the splenocytes were performed to assess leukocyte subpopulations. A. Representative FACS plots. Single live cells were gated for the CD45+ population and used for further analysis as indicated here. B. Results of FACS analysis. Percent of T cell, B cell, NK cell, monocyte, dendritic cell, granulocyte and macrophage in homozygous B-hCCR8 mice were similar to those in the C57BL/6 mice, demonstrating that CCR8 humanized does not change the overall development, differentiation or distribution of these cell types in spleen. Values are expressed as mean ± SEM.
-
Blood routine test

-
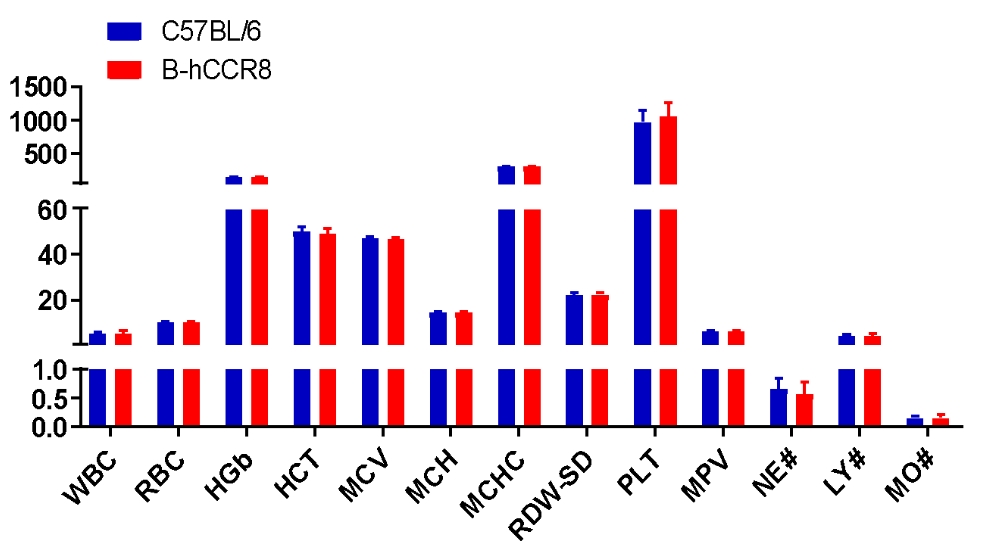
Complete blood count (CBC). Blood from female C57BL/6 and B-hCCR8 mice (n=8, 9 week-old) was collected and analyzed for CBC. The measurements of B-hCCR8 mice were similar to that in C57BL/6 mice, indicating that humanization does not change blood cell composition and morphology. Values are expressed as mean ± SEM.
-
Blood chemistry

-
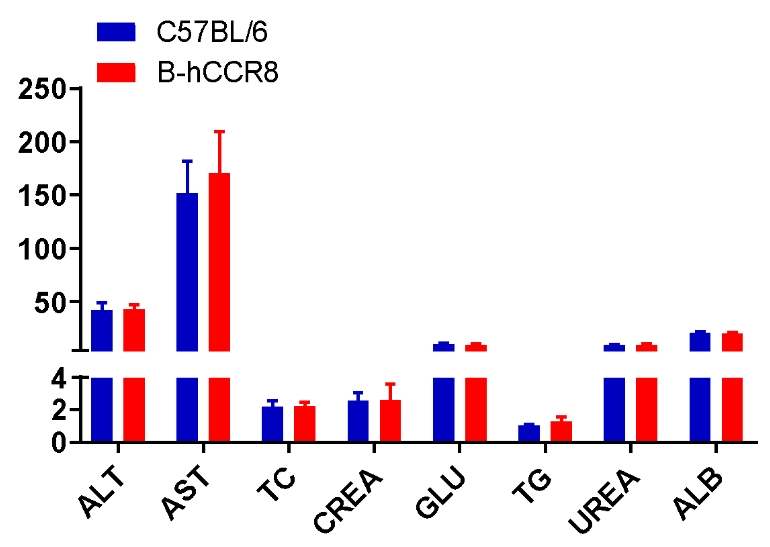
Blood chemistry tests of B-hCCR8 mice. Serum from the C57BL/6 and B-hCCR8 mice (n=8, 9 week-old) was collected and analyzed for levels of indicators. The measurements of B-hCCR8 mice were similar to that in C57BL/6 mice, indicating that humanization does not change the health of related tissues, such as liver. Values are expressed as mean ± SEM.
-
Summary for protein expression

-
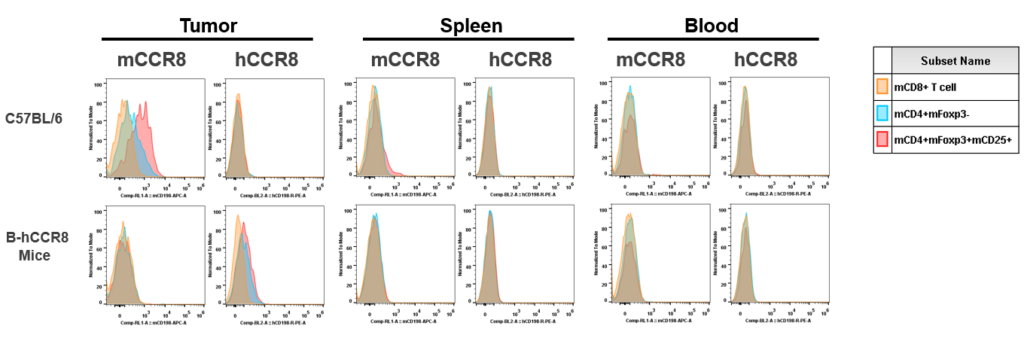
Strain specific analysis of CCR8 gene expression in B-hCCR8 mice by FACS. Human CCR8 was both detectable on CD4+ T cells and Treg cells in tumors of homozygous B-hCCR8 mice, but not in splenocytes and blood cells. Mouse CCR8 was detectable in tumors and slightly expressed in splenocytes of wild-type mice, but not in blood cells.
-
Leukocytes subpopulations in spleen

-
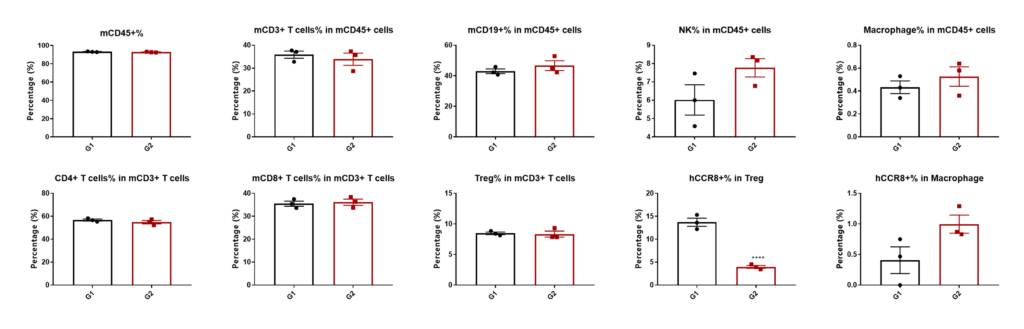
Analysis of leukocytes subpopulations in spleen by FACS.
Splenocytes were harvested at the endpoint of the experiment and flow cytometry analysis was performed to assess the leukocyte subpopulations. The percent of CCR8+ Treg cells were significant decrease in anti-human CCR8 antibody treatment group (G2). (*p<0.05, **p<0.01, ***p<0.001, ****p<0.0001)
-
In vivo efficacy of anti-human CCR8 antibody

-
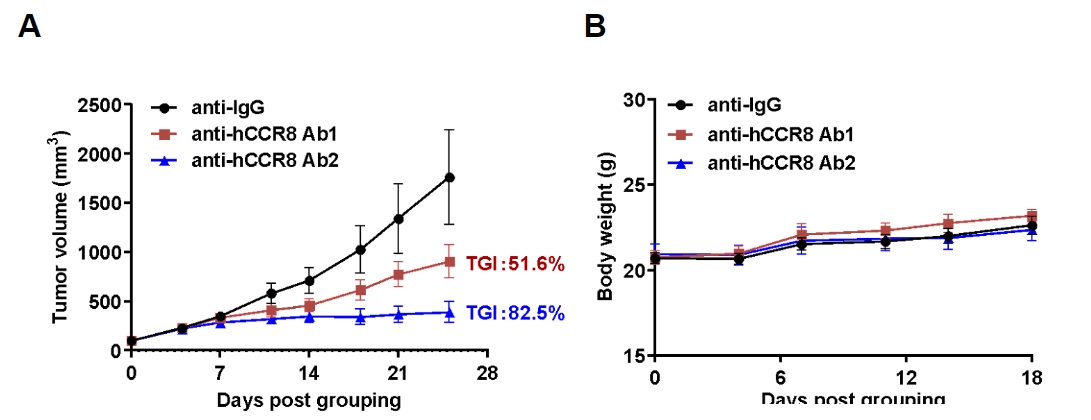
Antitumor activity of anti-human CCR8 antibody in B-hCCR8 mice bearing MC38 cells. Murine colon cancer MC38 cells were subcutaneously implanted into homozygous B-hCCR8 mice (female, 7 week-old, n=6). Mice were grouped when tumor volume reached approximately 100 mm3, at which time they were treated with anti-human CCR8 antibodies (in house). (A) Tumor growth curve. (B) Body weight changes during treatment. As shown, anti-human CCR8 antibodies were efficacious in controlling tumor growth in B-hCCR8 mice. B-hCCR8 mice provide a powerful preclinical model for in vivo evaluation of anti-human CCR8 antibodies. Values are expressed as mean ± SEM.
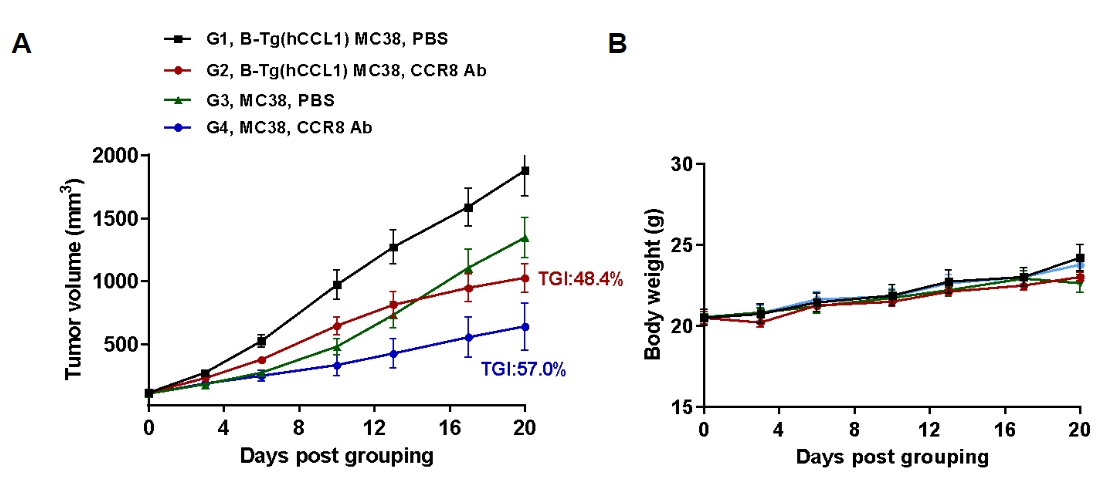
Antitumor activity of anti-human CCR8 antibody in B-hCCR8 mice. (A) Anti-human CCR8 antibody get from cooperation company inhibited MC38 tumor growth in B-hCCR8 mice. Murine colon cancer MC38 cells were subcutaneously implanted into homozygous B-hCCR8 mice (female, 8 week-old, n=7). Mice were grouped when tumor volume reached approximately 100 mm3, at which time they were treated with anti-human CCR8 antibody. (B) Body weight changes during treatment. As shown in panel A, anti-human CCR8 antibody were efficacious in controlling tumor growth in B-hCCR8 mice. B-hCCR8 mice provide a powerful preclinical model for in vivo evaluation of anti-human CCR8 antibody. Values are expressed as mean ± SEM.
-
Tumor infiltrates lymphocytes analysis

-
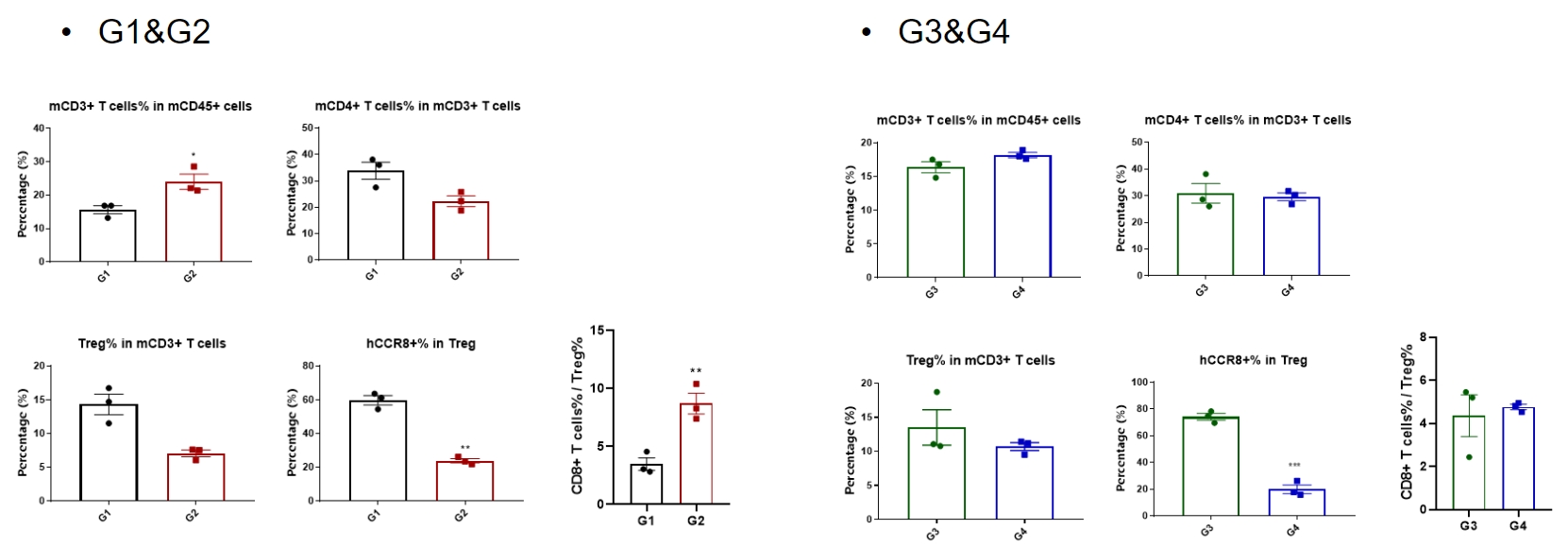
Analysis of tumor infiltrates lymphocytes by FACS.
TILs were harvested at the endpoint of the experiment and flow cytometry analysis was performed to assess the leukocyte subpopulations. The percent of CCR8+ Treg cells were significant decrease in anti-human CCR8 antibody treatment group (G2, G4), and the ratio CD8+ T cells to Treg cells increased significantly in G2, but not in G4. (*p<0.05, **p<0.01, ***p<0.001, ****p<0.0001)
-
Summary

-
- mRNA expression analysis:
Mouse Ccr8 mRNA was detectable in thymocyte of wild-type (+/+). Human CCR8 mRNA was detectable in B-hCCR8 mice (H/H) mice but not in wild-type mice.
- Protein expression analysis:
1) Human CCR8 was both detectable on CD4+ T cells and Treg cells in tumors of homozygous B-hCCR8 mice but not in wild-type mice, and mouse CCR8 was detectable only in wild-type mice.
2) Mouse CCR8 was slightly expressed in Tregs in spleen of wild-type mice, but human CCR8 was not detectable in homozygous B-hCCR8 mice. Because the CCR8 expression is low in the spleen, it is possible the CCR8 is expressed much lower in the spleen of homozygous B-hCCR8 mice such that we can’t detect it, or the antibody dose used in this experiment is not saturated.
- Leukocytes cell subpopulation analysis:
CCR8 humanized does not change the overall development, differentiation or distribution of immune cell types in spleen, thymus and blood.
- Complete blood count and blood chemistry:
The CBC of B-hCCR8 mice were similar to that in C57BL/6 mice, indicating that humanization does not change blood cell composition and morphology. In addition, the blood chemistry of B-hCCR8 mice was similar to that in C57BL/6 mice, indicating that humanization does not change the health of related tissues, such as liver.
- In vivo efficacy:
Anti-human CCR8 antibody was efficacious in controlling tumor growth in B-hCCR8 mice.


