C57BL/6-Cd274tm1(CD274)Bcgen Tnfrsf9tm1(TNFRSF9)Bcgen/Bcgen • 120535
| Product name | B-hPD-L1/h4-1BB mice |
|---|---|
| Catalog number | 120535 |
| Strain name | C57BL/6-Cd274tm1(CD274)Bcgen Tnfrsf9tm1(TNFRSF9)Bcgen/Bcgen |
| Strain background | C57BL/6 |
| NCBI gene ID | 3604,29126 (Human) |
| Aliases | ILA; 4-1BB; CD137; CDw137; IMD109; B7-H; B7H1; PDL1; PD-L1; ADMIO5; hPD-L1; PDCD1L1; PDCD1LG1 |
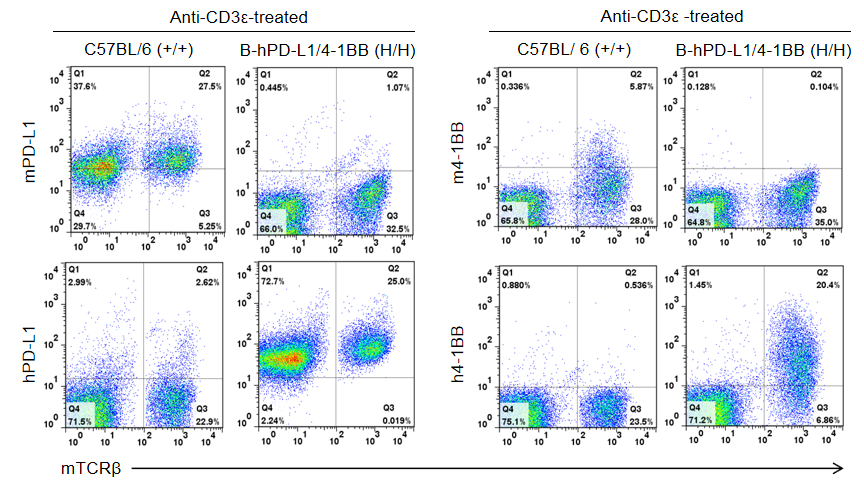
Strain specific PD-L1 and 4-1BB expression analysis in homozygous B-hPD-L1/h4-1BB mice by flow cytometry. Splenocytes were collected from wild type C57BL/6 mice (+/+) and homozygous B-hPD-L1/h4-1BB mice (H/H) stimulated with anti-CD3ε in vivo, and analyzed by flow cytometry with species-specific anti-PD-L1 and anti-4-1BB antibodies. Mouse PD-L1 and 4-1BB was detectable in C57BL/6 mice but not in homozygous B-hPD-L1/h4-1BB mice. Human PD-L1 and 4-1BB was exclusively detectable in homozygous B-hPD-L1/h4-1BB mice but not in C57BL/6 mice.
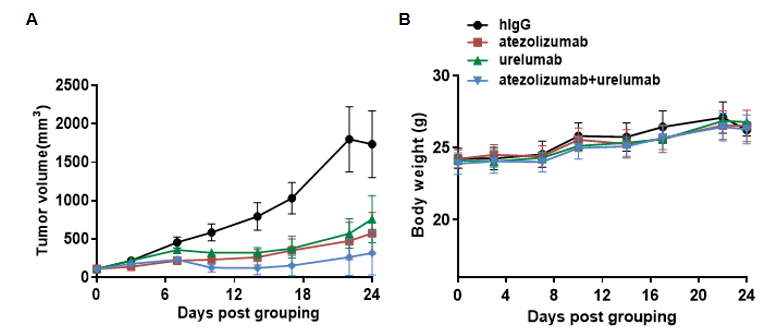
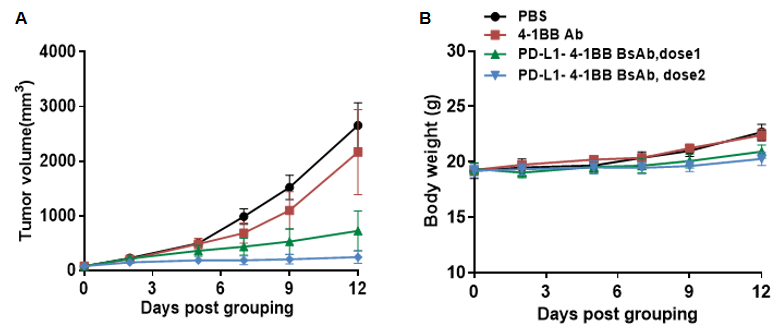
Antitumor activity of combination of anti-human PD-L1 antibody (atezolizumab,in house)and anti-human 4-1BB (urelumab, in house) in B-hPD-L1/h4-1BB mice. (A) combination of anti-human PD-L1 antibody and anti-human 4-1BB antibody inhibited MC38 tumor growth in B-hPD-L1/h4-1BB mice. Murine colon cancer MC38 cells were subcutaneously implanted into homozygous B-hPD-L1/h4-1BB mice (male, 6-week-old, n=6). Mice were grouped when tumor volume reached approximately 100 mm3, at which time they were treated with anti-human PD-L1 antibody and anti-human 4-1BB antibody with doses and schedules indicated in panel. (B) Body weight changes during treatment. As shown in panel A, combination of anti-human PD-L1 antibody and anti-human 4-1BB antibody were efficacious in controlling tumor growth in B-hPD-L1/h4-1BB mice, demonstrating that the B-hPD-L1/h4-1BB mice provide a powerful preclinical model for in vivo evaluation of combination of anti-human PD-L1 antibody and anti-human 4-1BB antibody. Values are expressed as mean ± SEM.