DSS IBD Model
Inflammatory bowel disease (IBD) refers to chronic inflammatory disease of the colon or gastrointestinal tract, including ulcerative colitis (UC) and Crohn’s disease (CD). Clinical symptoms of IBD include diarrhea, abdominal pain, intestinal bleeding/hematochezia, fever, and weight loss. The pathogenesis of IBD is not clear, however studies have shown that various factors such as genetic, immune system, external environment and intestinal microorganisms are associated with the occurrence of IBD.
A variety of preclinical mouse models are currently used to study IBD. Depending on the specific use, different induction methods can yield disease models. The mouse enteritis model conferred by dextran sulfate sodium (DSS) is the most widely used chemically-induced mouse IBD model. Acute ulcerative enteritis or chronic colitis is induced in mice by dissolving DSS in drinking water, resulting in loss of intestinal epithelial cells, cytokine release by non-specific immune cells, and disruption in the integrity of the mucosal barrier. IBD-induced mouse models experience significant weight loss, loose stools, hematochezia, and granulocyte infiltration, altogether suggesting that pathological features and clinical symptoms are similar to those seen in human ulcerative colitis.
Biocytogen generated a stable IBD-induced model using DSS in wild-type C57BL/6 mice, which can be used for preclinical studies and pharmacodynamic evaluation of inflammatory enteritis.
-
Establishment of an IBD Mouse Model Using DSS
-
Experimental mouse strains: C57BL/6, 7-8-week-old, female
Modeling reagent: Dextran Sulfate Sodium (DSS)
Modeling method: Supplement DSS in drinking water for 7 consecutive days
Clinical assessment: Disease activity index (DAI) score, including body weight and colon evaluation, and fecal morphology observation
Evaluation of UC: Colon weight and length
Pathological analysis: H&E staining
-
Induction of IBD in Mice
-
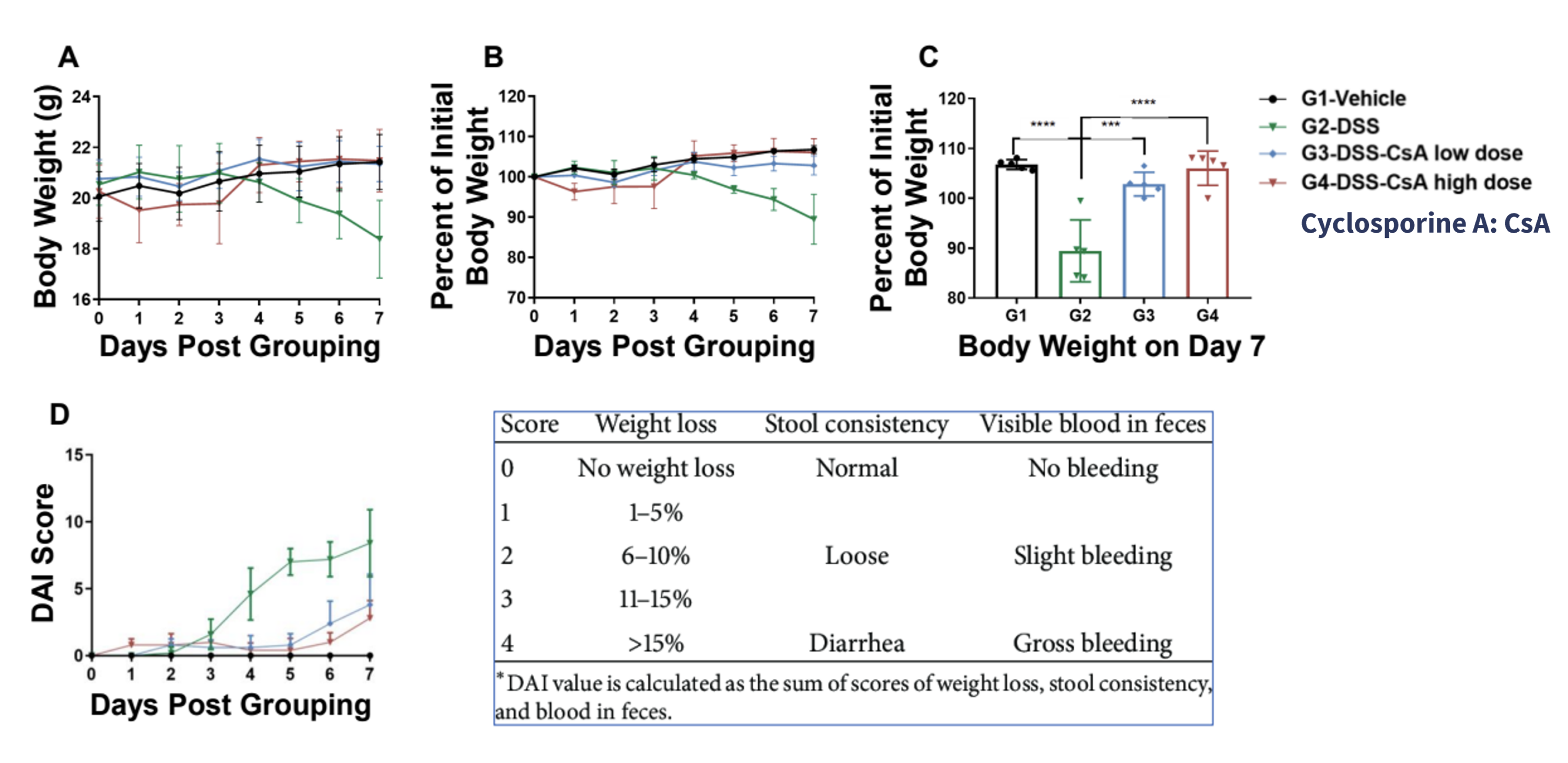
Wild-type C57BL/6 mice were provided drinking water containing DSS for 7 consecutive days for IBD induction. (A-C) Change in mouse body weight for each experimental group (G1-G4). (D) Disease activity index (DAI) score for each experimental group (G1-G4). Body weight of DSS-treated mice (G2) decreased compared to control vehicle-treated mice (G1), furthermore, the DAI score for DSS-treated mice indicated increased disease severity. Body weight and DAI scores for IBD mice treated with different doses of cyclosporin A (CsA) did not change significantly. Altogether, this data shows that IBD was successfully established in C57BL/6 mice by DSS, and CsA relieved clinical symptoms of IBD in a dose-dependent manner. Values are expressed as mean ± SEM. ***p<0.001, **** p<0.0001
-
Gross Sampling and Colon Evaluation
-
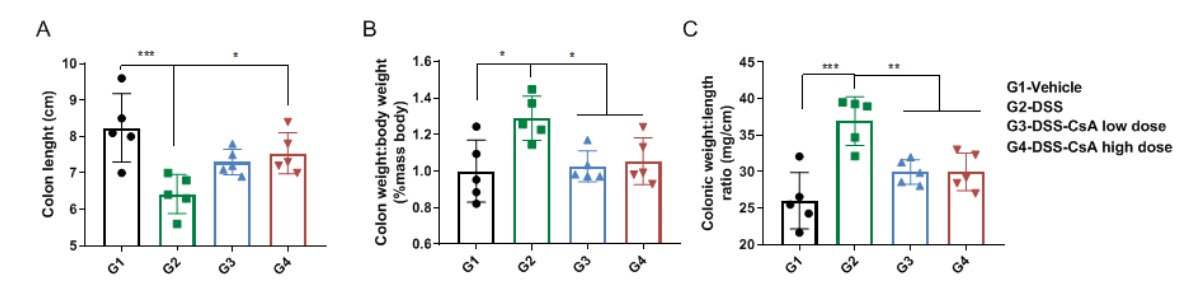
Comparison of colons in C57BL/6 mice with DSS-induced inflammatory bowel disease. (A) Colon length, (B) colon weight to body weight ratio and (C) the ratio of colon weight to colon length were measured for each experimental group (G1-G4). Colon weight and length were significantly abnormal in DSS-induced IBD mice (G2) compared to control vehicle-treated mice (G1). However, colon weight and length improved in CsA-treated mice (G3, G4), suggesting CsA alleviates colonic symptoms in IBD mice.
-
Pathological Assessment of CsA-Treated IBD Mice
-
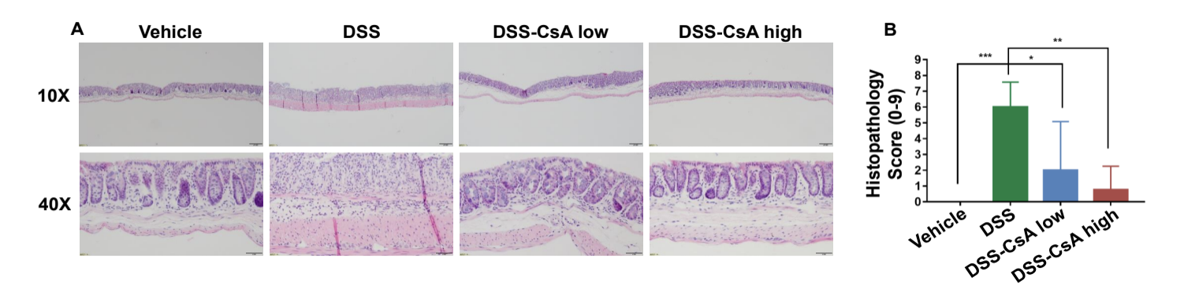
CsA treatment significantly alleviates IBD-like colon lesions in DSS-induced IBD mice. (A) H&E staining of colons and (B) pathological scores were assessed for each experimental group (G1-G4). Compared to control mice (G1), inflammatory cell infiltration was observed in DSS-treated mice (G2) indicative of destroyed intestinal epithelial cells and intestinal mucosal layer. In CsA-treated IBD mcie, colonic symptoms were significantly relieved in a dose-dependent manner.
Altogether, these data suggest that DSS colitis in C57BL/6 mice was established, and cyclosporin A (CsA) could significantly alleviate DSS-induced inflammatory bowel disease colon lesions.
-
Mouse Naïve T Cell Isolation and Adoptive Cell Transfer Workflow
-
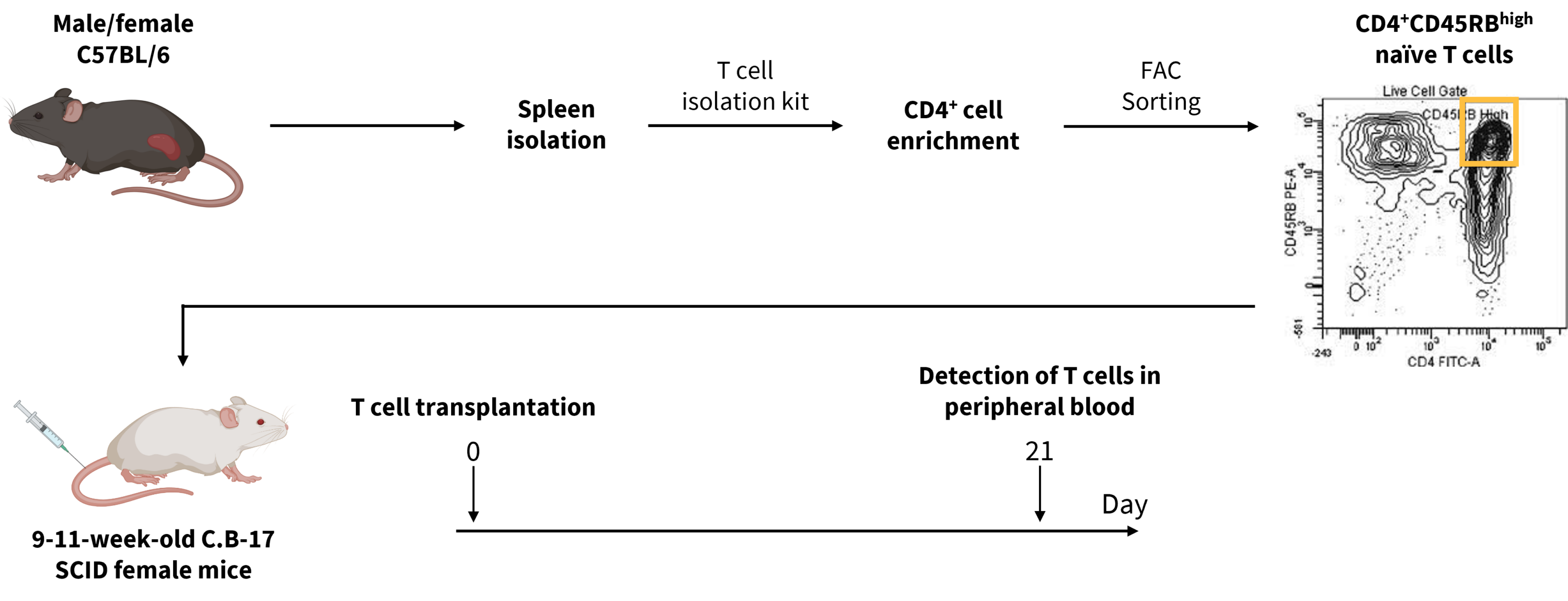
While DSS is widely used to recapitulate human IBD in mice, several preclinical models exist to study various aspects of disease onset and progression. In this pilot study, CD4+CD45RB high naïve T cells were isolated from male and female C57BL/6 mice and adoptively transferred into 9-11-week-old female SCID mice that lack T and B cells.
-
Induction of Inflammatory Bowel Disease by Adoptive T Cell Transfer
-
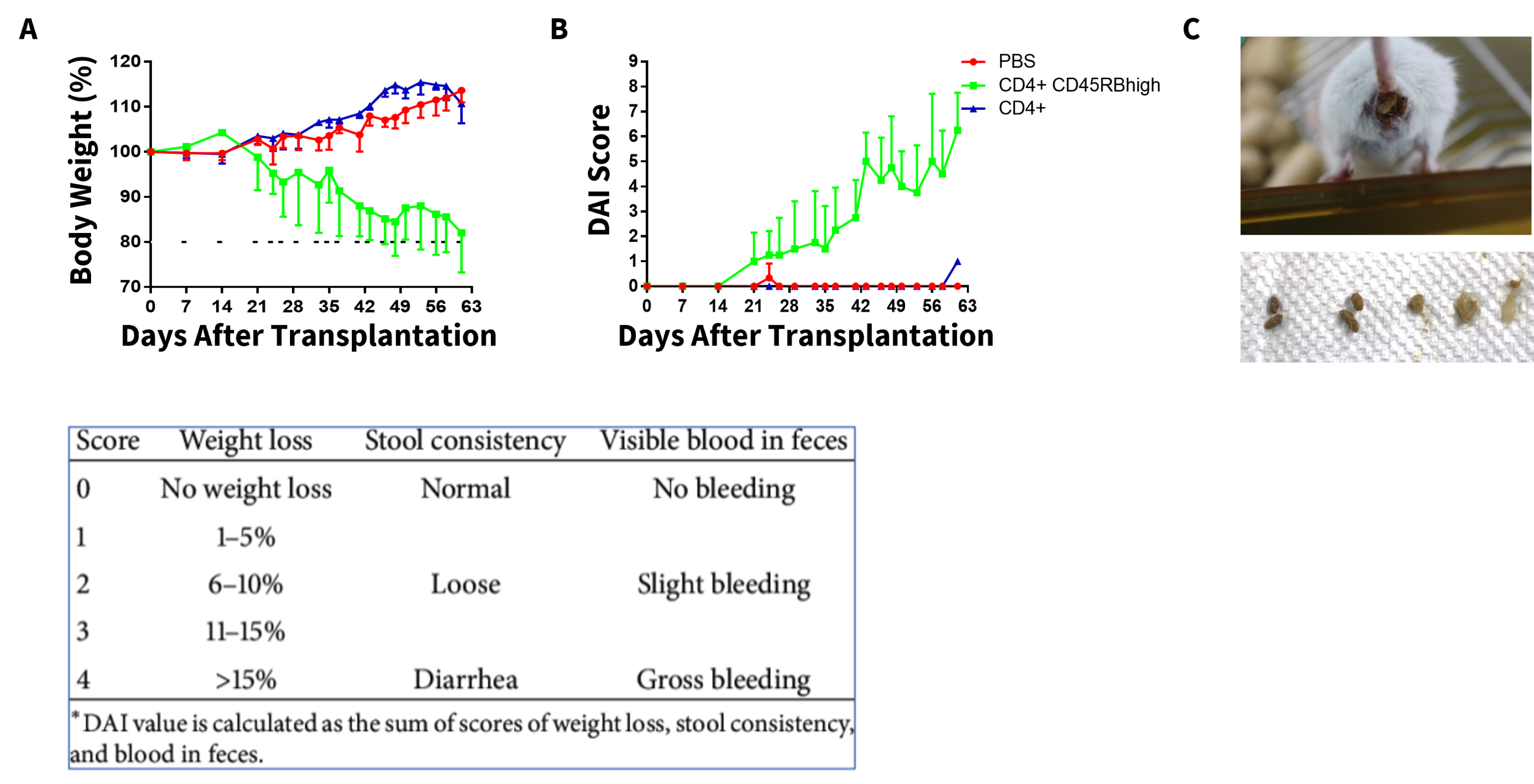
The CD4+CD45RB high population contains naïve T cells that are primed for activation, which can induce chronic small bowel and colon inflammation. Here, we see that adoptive T cell transfer-induced IBD was successfully established in SCID mice as shown by body weight loss (A), which correlated to an increase in disease activity index score (B), shown by the green line and altered stool consistency (C) compared with the PBS and CD4+ control groups.
-
Gross Sampling and Histologic Assessment of T Cell-Induced IBD Mice
-
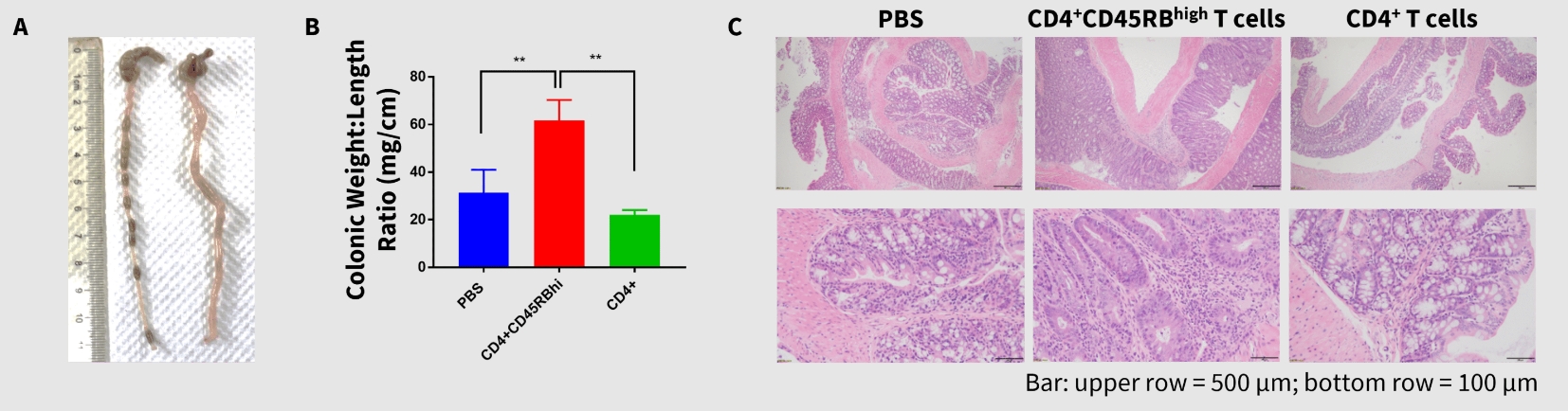
Gross anatomy and histological analysis of the colons in T cell-transplanted mice showed significantly disrupted colonic weight to length ratio (A, B) and morphology, such as hyperplasia and inflammatory cell infiltration (C). Together, these data indicate that in addition to DSS, CD4+CD45RB high adoptive T cell transfer can induce IBD in SCID mice, which recapitulates pathological features of human IBD.



