Experimental Autoimmune Encephalomyelitis (EAE) Model
Experimental Autoimmune Encephalomyelitis (EAE) is a relevant preclinical model of multiple sclerosis (MS). MS is a central nervous system chronic inflammatory disease which leads to brain inflammation and demyelination. MS is considered an autoimmune disorder caused by auto-reactive T cells with symptoms including muscle stiffness and paralysis, visual disturbances and blindness, sensory loss, and ataxia. The disease is prone to relapse and remission. There are several different animal models of MS. The EAE model is widely used because the pathological features of inflammation and demyelination induced in EAE is similar to that observed in MS disease. Among factors that influence the disease, the IL-23/IL-17 axis has been implicated in the pathogenesis of EAE and MS1-3.
The EAE model of multiple sclerosis can be induced by immunization with the proteins derived from the myelin sheath, such as myelin-oligodendrocyte glycoprotein (MOG), and Complete Freund’s Adjuvant (CFA) accompanied by an intraperitoneal injection of pertussis toxin (PTX) on the day of immunization and two days later. Myelin-specific T cells are activated in the periphery, migrate through the blood-brain barrier into the CNS and are reactivated, triggering a series of inflammatory reactions that lead to demyelination and axon cell death, and ultimately to nerve injury and disability.

Biocytogen provides a robust MOG-induced EAE model protocol for efficacy studies. Clinical symptoms are assessed using a standard scoring system that measures the degree of disease induction. Local demyelination and Inflammatory leukocyte infiltration can be visualized by histopathological staining. Furthermore, we can induce EAE in B-hIL17A mice, where the human IL-17A gene is knocked into the mouse IL-17A gene, thus providing a genetically humanized mouse EAE model of multiple sclerosis for convenient testing of novel therapeutics targeting human IL-17A.
Watch our webinar: Inflammatory Disease Modeling for Preclinical Studies
-
EAE Model Induction
-
Experimental mouse strains: C57BL/6, 9-10-week-old
Modeling reagent: Myelin oligodendrocyte glycoprotein (MOG) emulsion and pertussis toxin (PTX)
Modeling method: Immunized with MOG emulsion and PTX injection intraperitoneally
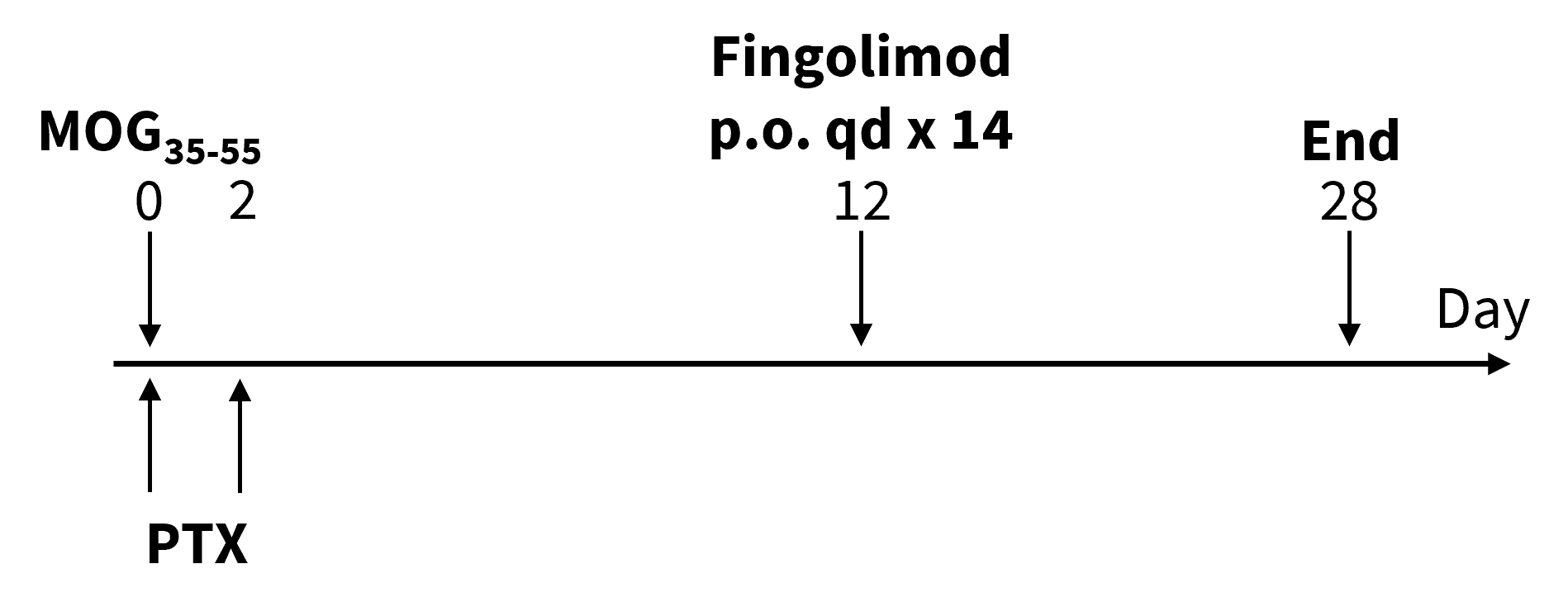
Experimental Autoimmune Encephalomyelitis (EAE) is an induced demyelinating disease model that closely resembles the progression and symptoms of the human neurological disease, Multiple Sclerosis (MS). Fingolimod was administered p.o. qd×14 beginning day 12 after MOG35-55 emulsion.
-
In Vivo Small Molecule Efficacy in EAE Mice
-
In vivo small molecule efficacy in wild-type EAE mice induced by MOG35-55 emulsion and PTX.
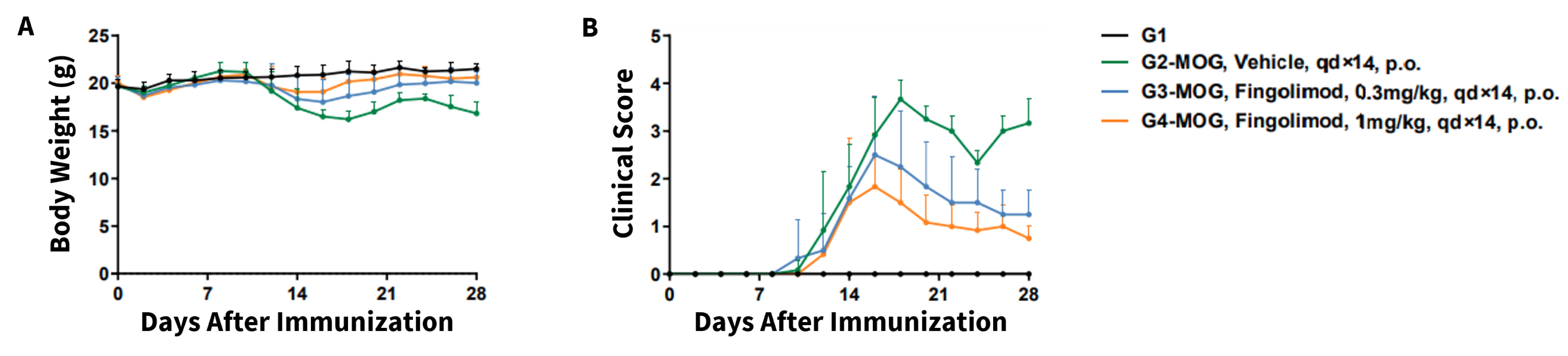
In vivo small molecule efficacy in wild-type EAE mice induced by MOG35-55 emulsion and PTX. (A) Body weight, and (B) clinical scores were measured in 10-week-old wild-type C57BL/6 mice (n=6) exposed to vehicle control (G1) MOG35-55 + Vehicle (G2) or MOG35-55 + Fingolimod (G3,G4) as indicated in the dosing regimen. Compared to vehicle control (G1), MOG35-55 treated mice (G2-G4) displayed tail weakness, lameness, hind limb paralysis and other symptoms, resulting in an increased clinical score. This data demonstrates successful EAE induction in wild-type C57BL/6 mice. Clinical symptoms were reduced in a dose-dependent manner when Fingolimod was administered. Values are expressed as mean ± SEM. MOG35-55: Myelin oligodendrocyte glycoprotein polypeptide 35 and 55. PTX: pertussis toxin.
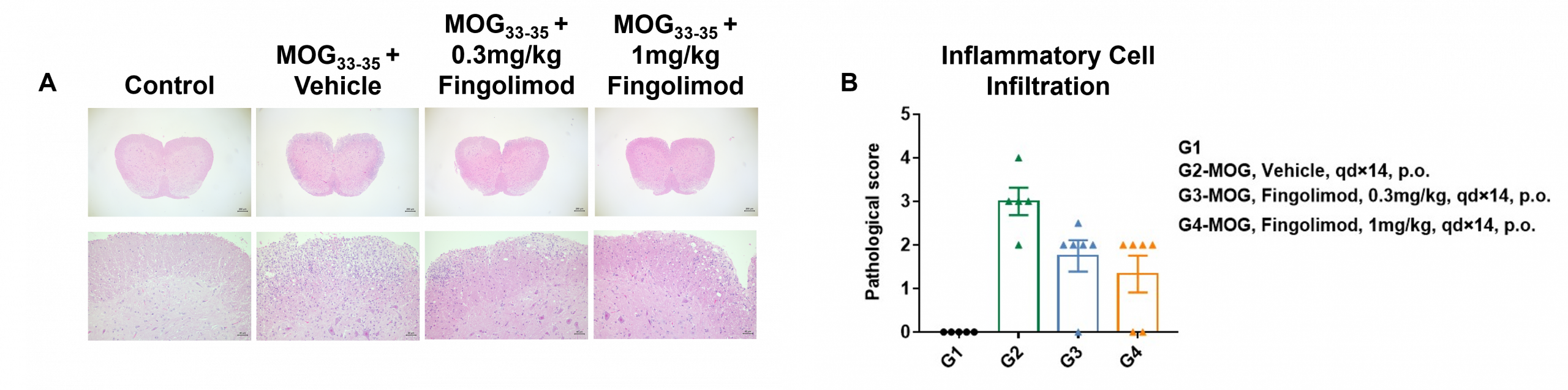
Local inflammatory responses in the central nervous system (CNS) of EAE mice. Spinal cord tissue sections were isolated from wild-type C57BL/6 mice 28 days after MOG35-55 immunization and analyzed by hematoxylin and eosin (H&E) staining (A) to score infiltrating inflammatory cells (B). Compared to control-treated wild-type mice (G1), inflammatory cell infiltration increased in wild-type mice immunized with MOG35-55 (G2), demonstrating successful EAE induction. Inflammatory cell infiltration reduced in wild-type EAE-induced mice treated with Fingolimod (G3, G4). Values are expressed as mean ± SEM.



