Basic Information
Description
Human HSC (hematopoietic stem cells ) engrafted humanized mice are a powerful model that allows researchers to examine the human immune system and related primates’ alien virus research and cellular immunotherapy preclinical evaluation. Although B-NDG humanized mice support the investigation of human immunity in vivo, a species barrier between human immune cells and the mouse microenvironment limits human adaptive and innate immune function. Such as frequencies of human NK cells did not reach physiological levels in B-NDG humanized mice.
-
Targeting strategy

-
Gene targeting strategy for B-NDG hIL15 mice. The CDS region of the human IL15 gene was inserted after the 5′UTR of the mouse Il15 gene.
-
mRNA expression analysis

-
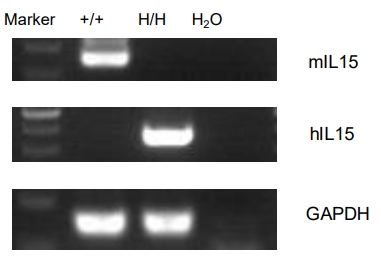
Strain specific analysis of IL15 gene expression in wild-type B-NDG mice and homozygous B-NDG hIL15 mice by RT-PCR. Mouse Il15 mRNA was detectable only in splenocytes of wild-type B-NDG mice (+/+). Human IL15 mRNA was detectable only in homozygous B-NDG hIL15 mice (H/H), but not in B-NDG mice.
-
Protein expression analysis

-
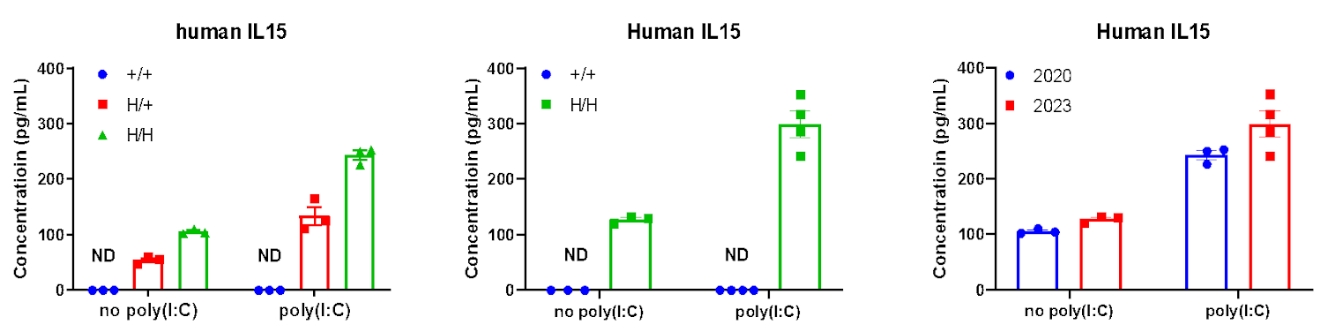
Strain specific IL15 expression analysis in wild-type B-NDG mice and heterozygous/homozygous B-NDG hIL15 mice by ELISA. Serum was collected from wild-type B-NDG mice (+/+), heterozygous B-NDG hIL15 mice (H/+) and homozygous B-NDG hIL15 mice (H/H) stimulated with or without poly(I:C) in vivo, and analyzed by ELISA with species-specific IL15 ELISA kit. Human IL15 was detectable in heterozygous and homozygous B-NDG hIL15 mice but not in B-NDG mice (male, 6-week-old, detected in 2020.09). In 2023.07, Human IL15 was re-detected in B-NDG mice and B-NDG hIL15 mice. The results showed that the expression level of human IL15 in the serum of B-NDG hIL15 mice was similar to that detected in 2020.
-
Hematology analysis

-
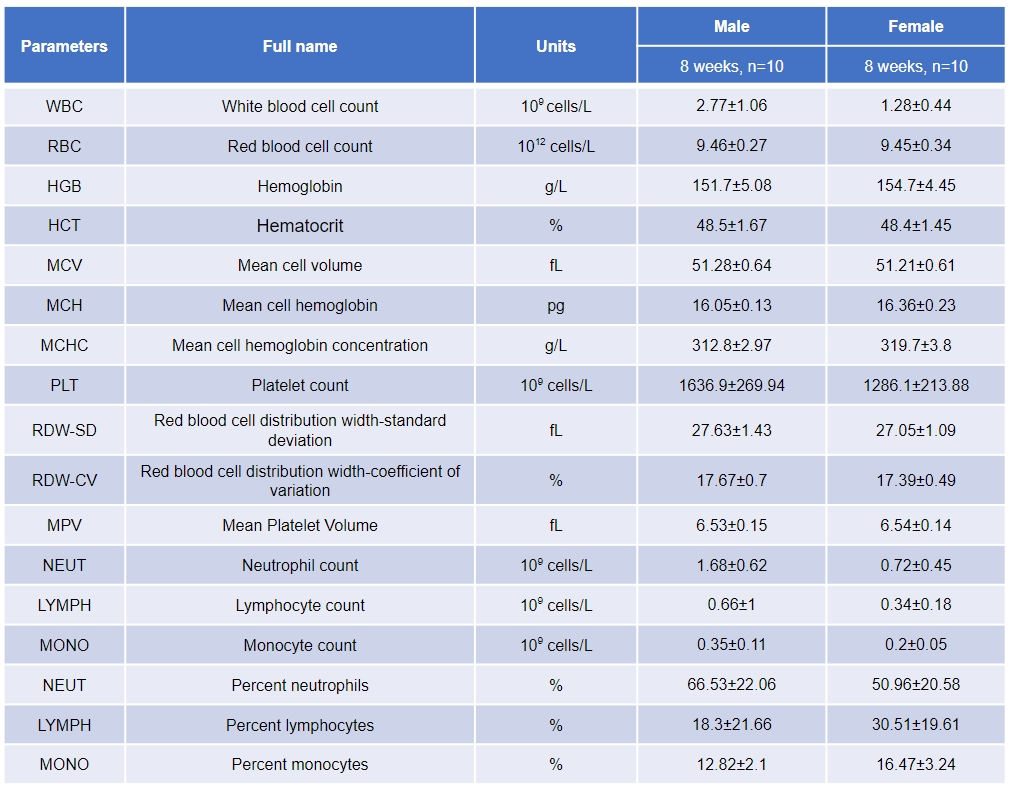
Complete blood count (CBC) of B-NDG hIL15 mice. Values are expressed as mean ± SD.
-
Engraftment of human CD34+ HSCs in B-NDG hIL15 mice to reconstitute human immune system (adult mice)

-
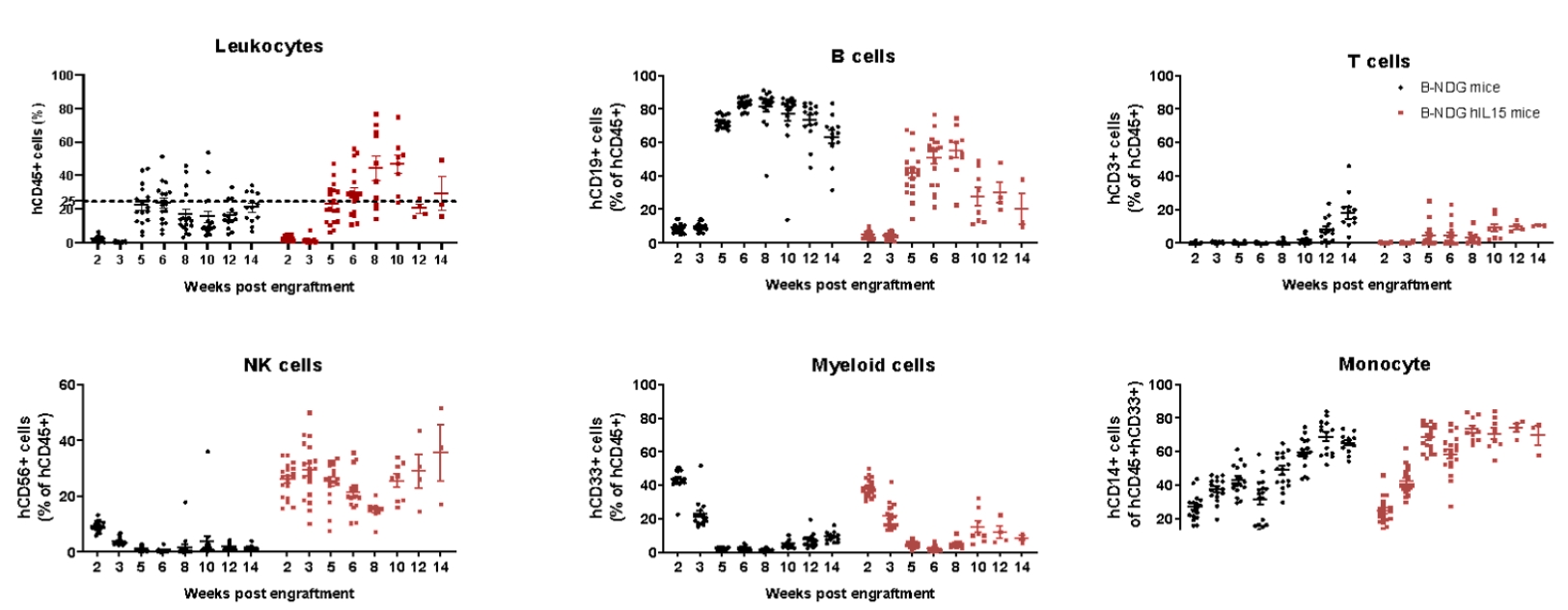
Human immune cell phenotyping in B-NDG hIL15 engrafted with human HSCs. Human CD34+ cells (0.15 M) were intravenously injected into homozygote B-NDG hIL15 (female, 6 week-old, n=19) and B-NDG mice (female, 6 week-old, n=17). All mice were treated with 1.6 Gy irradiation. Representative flow cytometric analysis of peripheral blood lymphocyte from mice after engraftment with human CD34+ cells. B-NDG hIL15 show a higher percentage of human NK cells compared with B-NDG. Our results suggest that human NK, T, and B cells in reconstituted B-NDG IL15 mice were successfully propagated.
-
CDX model of solid tumor established with huHSC-B-NDG hIL15 mice (adult mice)

-
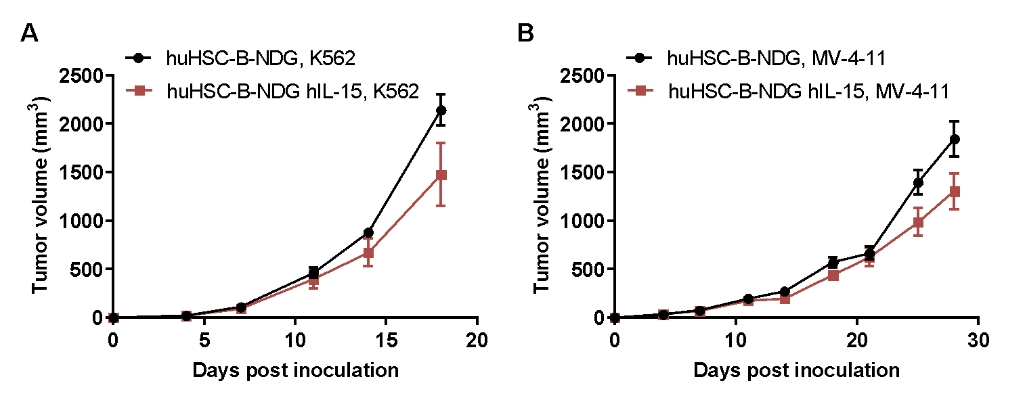
CDX model were well established on human immune system engraftment mice. Human CD34+ cells(0.15M) were intravenous implanted into homozygote B-NDG hIL15 (female, 7 week-old, n=30) and B-NDG mice (female, 7 week-old, n=30). All mice were treated with 1.6gry-irradiation. Mice were grouped as hCD45% ≥ 10%. K562 cells(1E6), MV-4-11(1E7), Panc-1(5E6) were subcutaneously implanted into B-NDG hIL15 and B-NDG mice respectively. (A-C) The mean ± SEM of tumor sizes. K562 and MV-4-11 were successfully tumorigenic on a mouse model of human HSC immune system reconstitution, and tumor growth was delayed in B-NDG hIL15 mice compared to B-NDG mice.
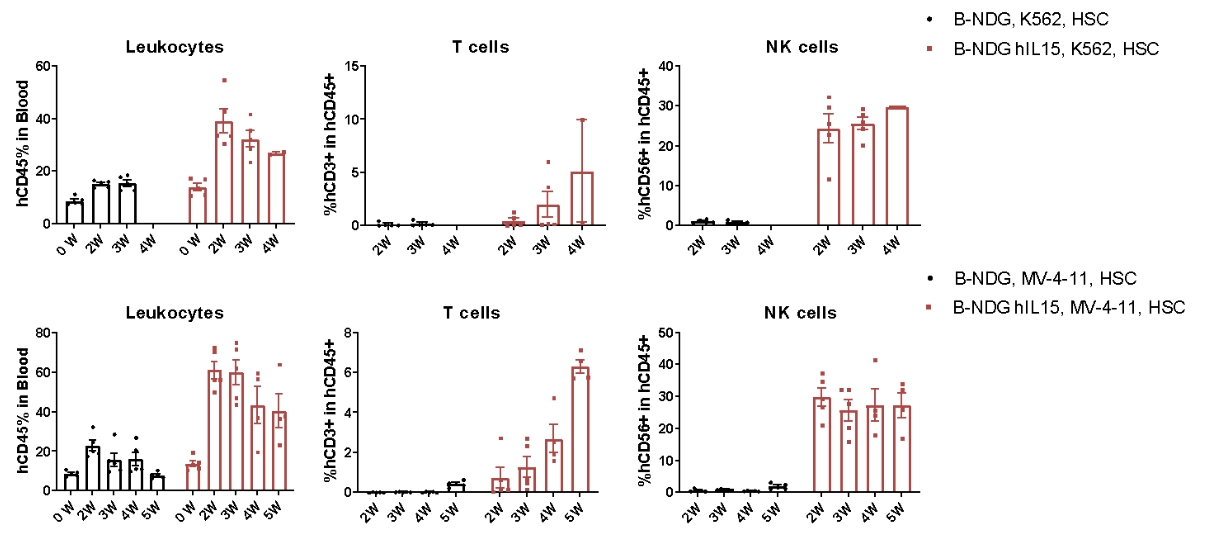
CDX model were well established on human immune system engraftment mice. Human CD34+ cells(0.15M) were intravenous implanted into homozygote B-NDG hIL15 (female, 7 week-old, n=30) and B-NDG mice (female, 7 week-old, n=30). All mice were treated with 1.6gry-irradiation. Mice were grouped as hCD45% ≥ 10%. K562 cells(1E6), MV-4-11 (1E7) were subcutaneously implanted into B-NDG hIL15 and B-NDG mice respectively. During the establishment of CDX model using K562 cells and MV-4-11, the proportion of immune cells in the blood of mice were measured. The results showed that during the establishment of tumor models, the proportion of leukocytes, T cells and NK cells in the blood of B-NDG hIL15 mice was higher than B-NDG mice.
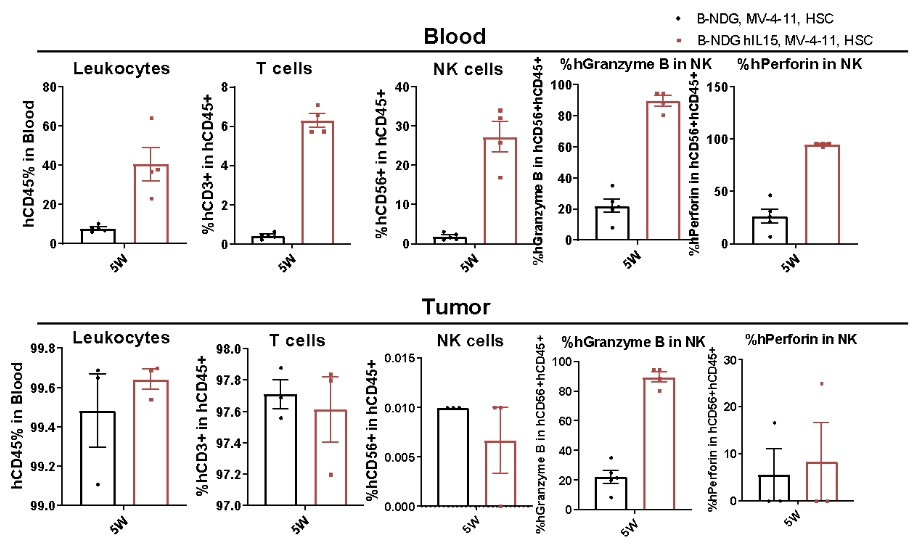
CDX model were well established on human immune system engraftment mice Human CD34+ cells(0.15M) were intravenous implanted into homozygote B-NDG hIL15 and B-NDG mice. All mice were treated with 1.6Gy-irradiation. Mice were grouped as hCD45% ≥ 10%. MV-4-11(1E7) were subcutaneously implanted into B-NDG hIL15 and B-NDG mice respectively. The proportion and function of NK cells in mouse blood and tumor tissues were measured at the end of the experiment. The results showed that B-NDG hIL15 show a higher percentage of human NK cells compared with B-NDG, and NK cell express functional proteins.
-
CDX model of lymphoma established in huHSC-B-NDG hIL15 mice (adult mice)

-
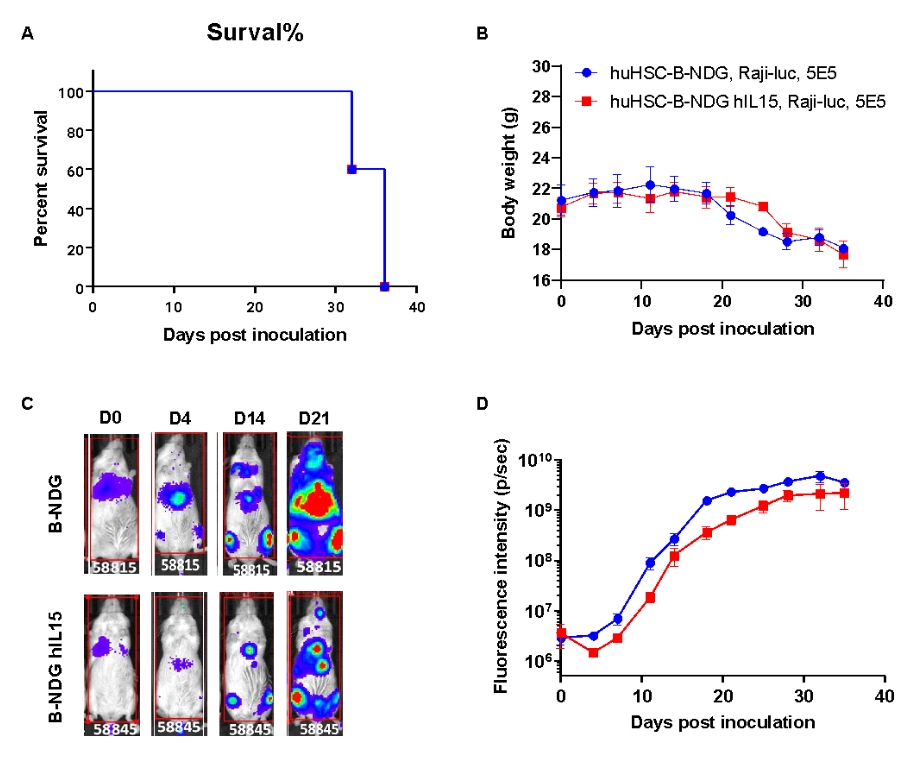
Raji lymphoma CDX model was well established in huHSC-B-NDG hIL15 mice. Human CD34+ HSCs (0.15 M) were intravenously engrafted into B-NDG mice and B-NDG hIL15 mice within 4-12 hours after being irradiated with 1.6 Gy of X-ray. Mice were grouped when the proportion of hCD45+ cells in blood was more than 10%, at which time B-luciferase-GFP Raji cells (5E5) were intravenously inoculated into B-NDG mice and B-NDG hIL15 mice respectively. (A) Survival curve; (B) Changes of body weight; (C) Imaging of mice to observe tumor growth; (D) Fluorescence intensity curve of tumor cells. The results showed that B-luciferase-GFP Raji cells were successfully tumorigenic in huHSC-B-NDG hIL15 mice. The tumor growth in huHSC-B-NDG hIL15 mice was slightly shower than that in huHSC-B-NDG mice.
-
Engraftment of human CD34+ HSCs in B-NDG hIL15 mice enhances reconstitution of human NK cells (neonatal mice)

-
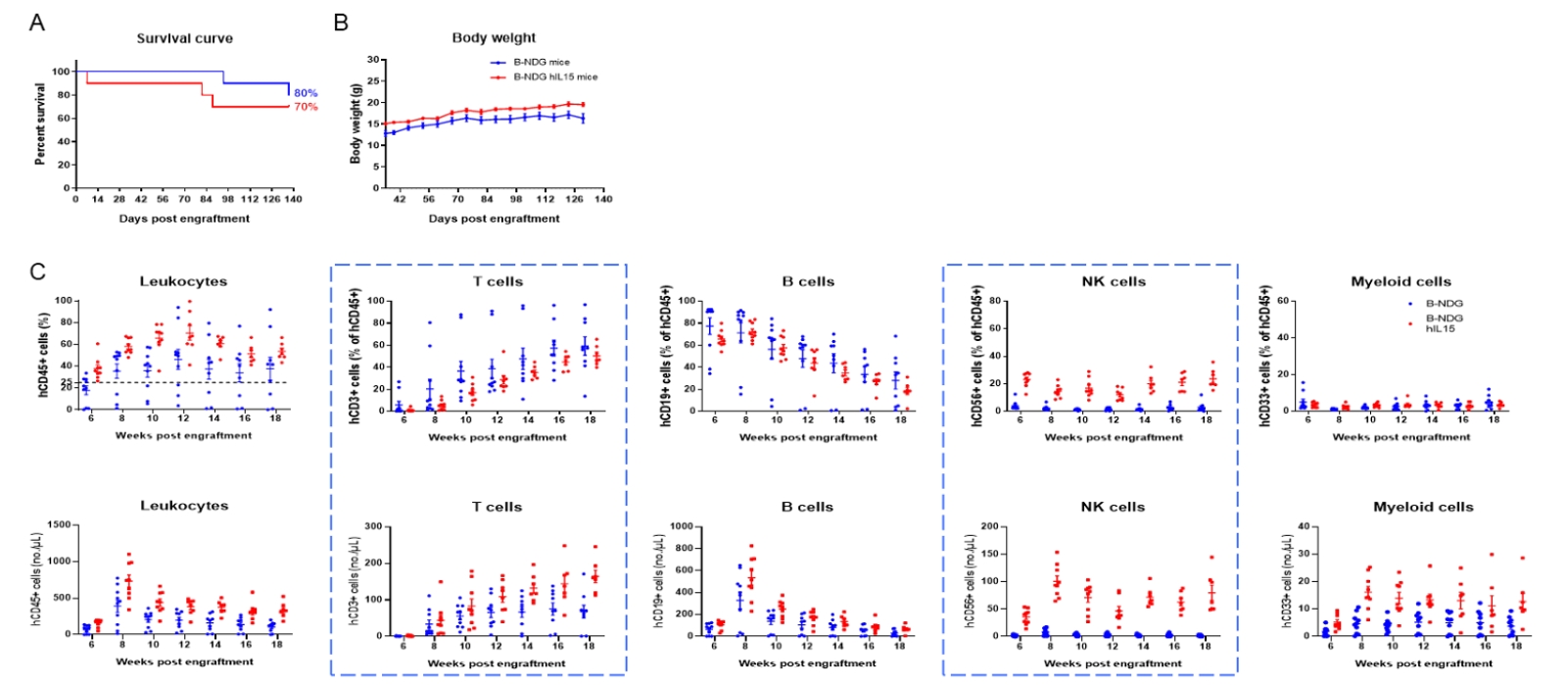
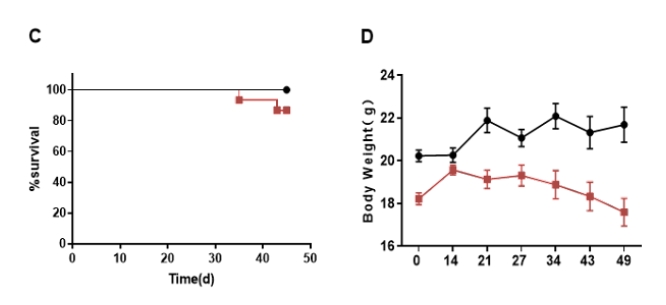
Engraftment of human CD34+ HSCs in neonatal B-NDG hIL15 mice successfully reconstituted human T, B and NK cells, a small number of myeloid cells. Human CD34+ HSCs (3E4) were engrafted via the facial vein of B-NDG hIL15 mice and B-NDG mice (female, 24-48 hour-old, n=10) within 4-12 hours after being irradiated with 0.9 or 1.0 Gy of X-ray. The reconstitution level of human immune cells in peripheral blood was analyzed by flow cytometry. (A) Survival curve of human CD34+ HSCs engrafted mice; (B) Body weight; (C) Percentages and number of reconstituted human immune cells. The results showed that the survival rate of huHSC-B-NDG hIL15 mice reached to 70% at 20 weeks after HSCs engraftment. But the lived mice continued to gain body weight. The percentages of human CD45+ cells and NK cells in huHSC-B-NDG hIL15 mice were much higher than that in huHSC-B-NDG mice. But the number of all cell types including human CD45+ cells, T cells, B cells, NK cells and myeloid cells in huHSC-B-NDG hIL15 mice were much more than that in huHSC-B-NDG mice.
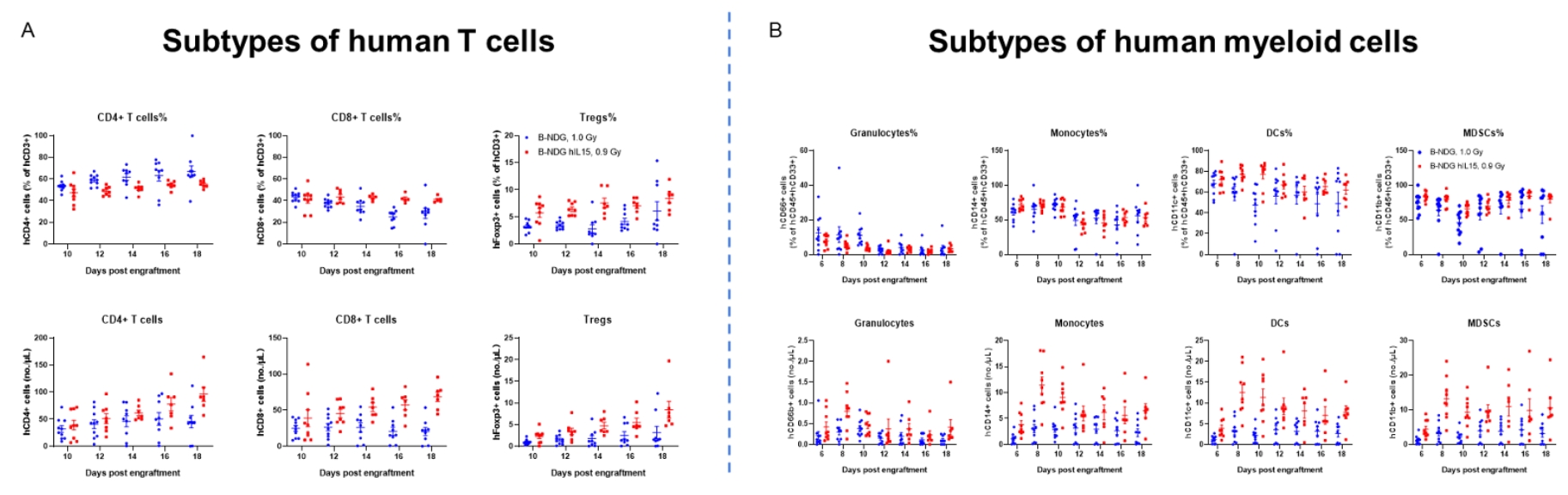
Engraftment of human CD34+ HSCs in neonatal B-NDG hIL15 mice successfully reconstituted human T, B and NK cells, a small number of myeloid cells. Human CD34+ HSCs (3E4) were engrafted via the facial vein of B-NDG hIL15 mice and B-NDG mice (female, 24-48 hour-old, n=10) within 4-12 hours after being irradiated with 0.9 or 1.0 Gy of X-ray. The reconstitution level of human immune cells in peripheral blood was analyzed by flow cytometry. (A) Percentages and number of reconstituted human T cells. (B) Percentages and number of reconstituted human myeloid cells. The results showed that the percentages of human CD8+ T cells, Tregs, monocytes, DCs and MDSCs in huHSC-B-NDG hIL15 mice were higher than that in huHSC-B-NDG mice. But the number of all cell types including human CD4+ T cells, CD8+ T cells, Tregs, granulocytes, monocytes, DCs and MDSCs in huHSC-B-NDG hIL15 mice were much more than that in huHSC-B-NDG mice.

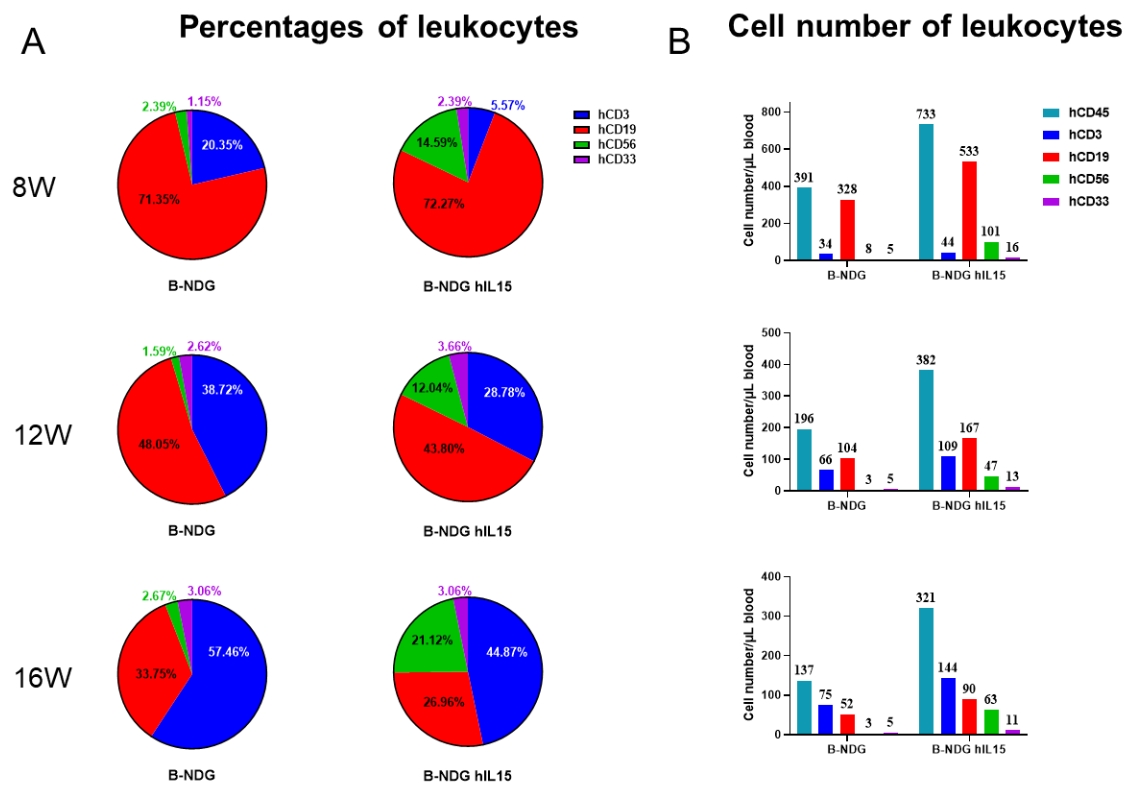
Engraftment of human CD34+ HSCs in neonatal B-NDG hIL15 mice successfully reconstituted human T, B and NK cells, a small number of myeloid cells. Human CD34+ HSCs (3E4) were engrafted via the facial vein of B-NDG hIL15 mice and B-NDG mice (female, 24-48 hour-old, n=10) within 4-12 hours after being irradiated with 0.9 or 1.0 Gy of X-ray. The reconstitution level of human immune cells in peripheral blood was analyzed by flow cytometry. (A, B) Statistical analysis of the percentages and number of reconstituted human immune cells. The results showed that reconstitution level of human NK cells in huHSC-B-NDG hIL15 mice was much higher than that in huHSC-B-NDG mice. Meanwhile, the reconstitution levels of human T, B and myeloid cells were also increased in huHSC-B-NDG hIL15 mice compared to that in huHSC-B-NDG mice.
-
In vitro cytotoxicity assay of isolated human NK cells from huHSC-B-NDG hIL15 mice

-
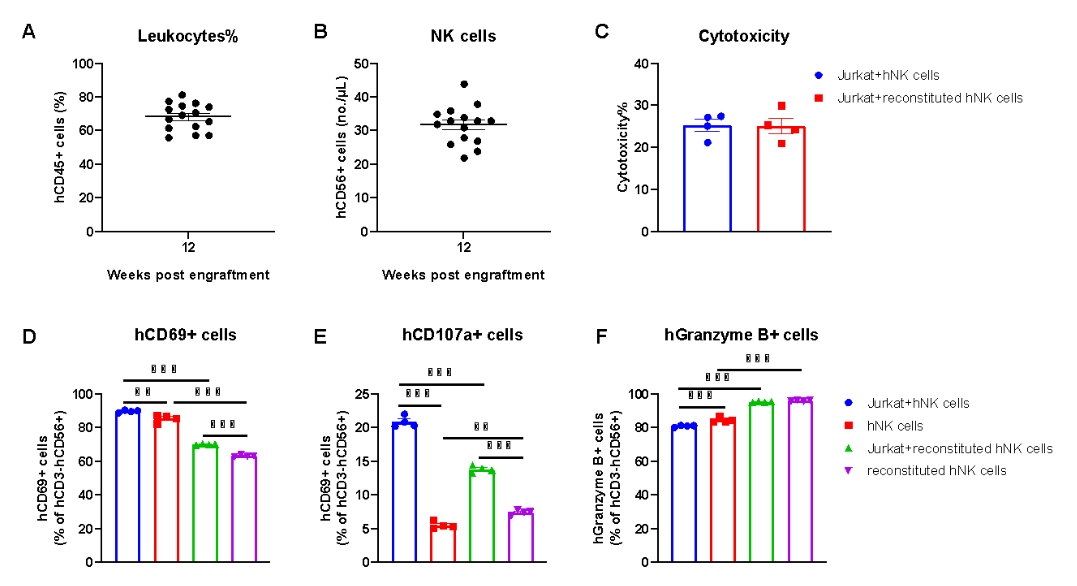
NK cells isolated from the spleen of huHSC-B-NDG hIL15 mice possess tumor-killing activity against Jurkat cells. NK cells were isolated from the spleen of huHSC-B-NDG hIL15 mice 14 weeks after hCD34+ HSCs engraftment (n=4). The isolated NK cells were cultured with human T lymphoblastoid cell line Jurkat cells and the cytotoxicity were measured with LDH assay. Human NK cells isolated from PBMCs were used as positive control. (A) The proportion of reconstituted human CD45+ cells ranged from 56% to 81%. (B) The number of reconstituted human NK cells ranged from 22 to 44 per microliter of blood. (C) There was no significant difference in cytotoxicity of reconstituted human NK cells compared to human PBMC-purified NK cells. (D-F) Activity markers of NK cells were detected with flow cytometry. Different proportions of NK cells expressing hCD69, hCD107a and hGranzyme B could be detected in reconstituted NK cells. After incubation with Jurkat cells, the proportion of NK cells expressing hCD69 and hCD107a increased; There was no significant change in the proportion of NK cells expressing hGranzyme B. Compared with PBMC-purified NK cells, the proportion of NK cells expressing hCD69 and hCD107a in reconstituted NK cells was lower; But the proportion of cells expressing hGranzyme B was higher. The results indicated that the reconstituted human NK cells in huHSC-B-NDG hIL15 mice possess similar cytotoxicity compared to human NK cells purified from PBMCs.
-
In vivo efficacy of anti-human CLDN18.2 antibody in CDX model established with huHSC-B-NDG hIL15 mice (adult mice)

-
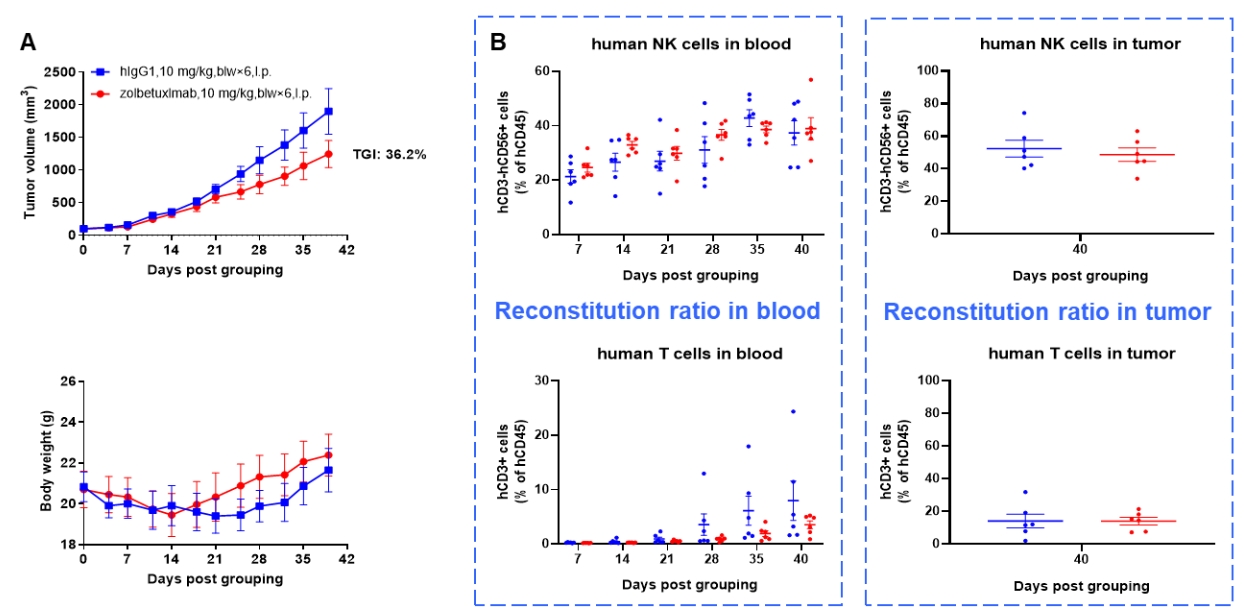
Antitumor activity of anti-human CLDN18.2 antibody in lung cancer cell line A549 CDX model established with huHSC-B-NDG hIL15 mice. Human CD34+ HSCs (0.15 M) were intravenously engrafted into B-NDG hIL15 mice within 4-12 hours after being irradiated with 1.6 Gy of X-ray. B-hCLDN18.2 A549 cells (1E7) were subcutaneously inoculated into B-NDG hIL15 mice when the proportion of hCD45+ cells in blood was more than 25%. Mice were grouped when the tumor volume reached 80-100 mm3 at which time they was treated with anti-human CLDN18.2 antibody (zolbetuximab, in house) with dose and schedule indicated in panel (female, 6-week-old, n=6). Peripheral blood was collected weekly to analyze the reconstitution levels of human NK cells and T cells. Tumor tissues were taken at the end of the experiment to detect the level of infiltrating human NK cells and T cells. The results showed that zolbetuximab could effectively inhibit tumor growth in huHSC-B-NDG hIL15 mice. High levels of human NK cells could be detected in both peripheral blood and tumor tissues. Human T cells could also be detected in peripheral blood and tumor tissues.
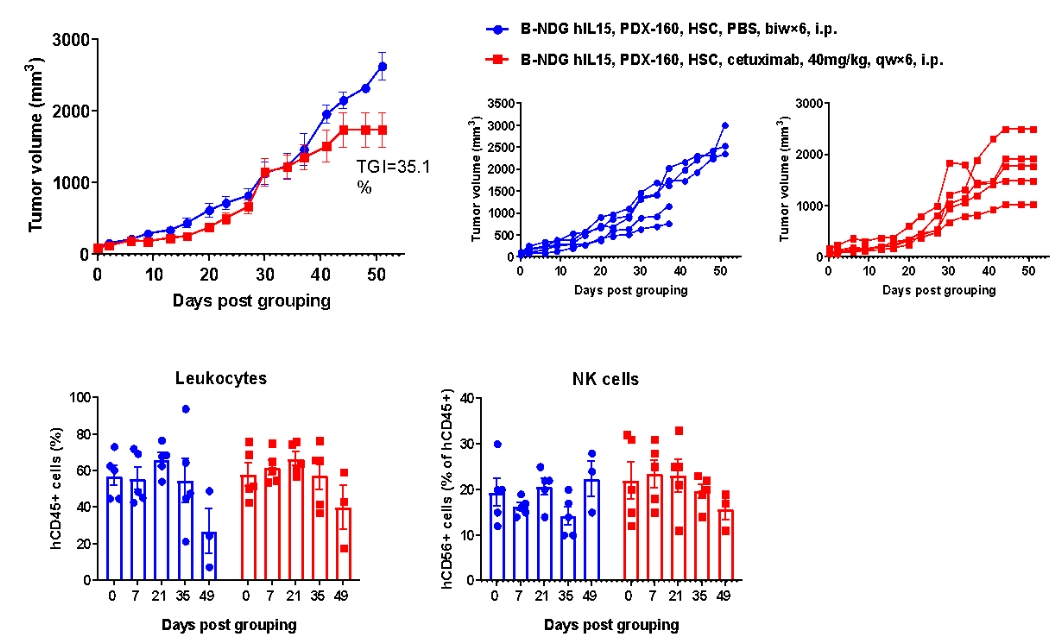
Antitumor activity of anti-human EGFR antibody in pancreatic cancer PDX model established with huHSC-B-NDG hIL15 mice. Human CD34+ HSCs (0.15 M) were intravenously engrafted into B-NDG hIL15 mice within 4-12 hours after being irradiated with 1.6 Gy of X-ray. Pancreatic cancer PDX (BP0160-R4P7) were inoculated into B-NDG hIL15 mice. Mice were grouped when the tumor volume reached approximately 100 mm3, at which time they were treated with anti-human EGFR antibody (cetuximab, in house) with dose and schedule indicated in panel. The proportion of leukocytes and NK cells in blood were measured once every other week. The results showed that cetuximab could effectively inhibit tumor growth in pancreatic cancer PDX model established with huHSC-B-NDG hIL15 mice. High levels of human NK cells could be detected in peripheral blood.
-
Engraftment of human NK cells purified from hPBMCs in B-NDG hIL15 mice enhances reconstitution of human NK cells

-
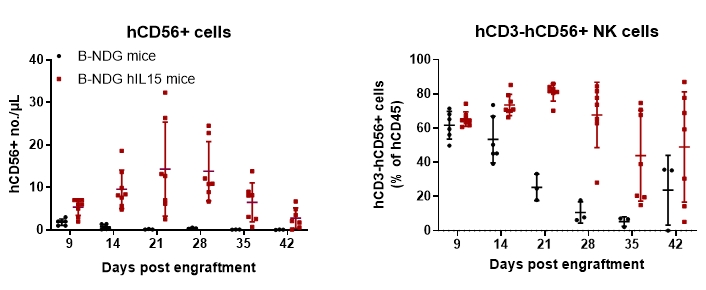
Engraftment of human NK cells in B-NDG hIL15 mice enhanced the reconstitution of human NK cells. Female 6-week-old of B-NDG mice (n = 7) and B-NDG hIL15 mice (n = 8) were irradiated with 1.2 Gy, and NK cells (2E6) obtained after purification of human PBMCs were intravenously injected. Peripheral blood was taken weekly to analyze the reconstitution level of human immune cells. The results showed that the proportion of NK cells obtained from PBMC purified reached 80%. Compared with B-NDG mice, the number and proportion of reconstituted human NK cells (CD3-CD56+) were significantly higher in B-NDG hIL15 mice. The proportion of NK cells remained around 80% after 2 weeks of reconstitution and a higher proportion of NK cells remained after 6 weeks of reconstitution. Reconstituted human NK cells were predominantly CD16+ NK cells with cytotoxic activity.
-
Expression analysis of functional markers and surface receptors on reconstituted human NK cells

-
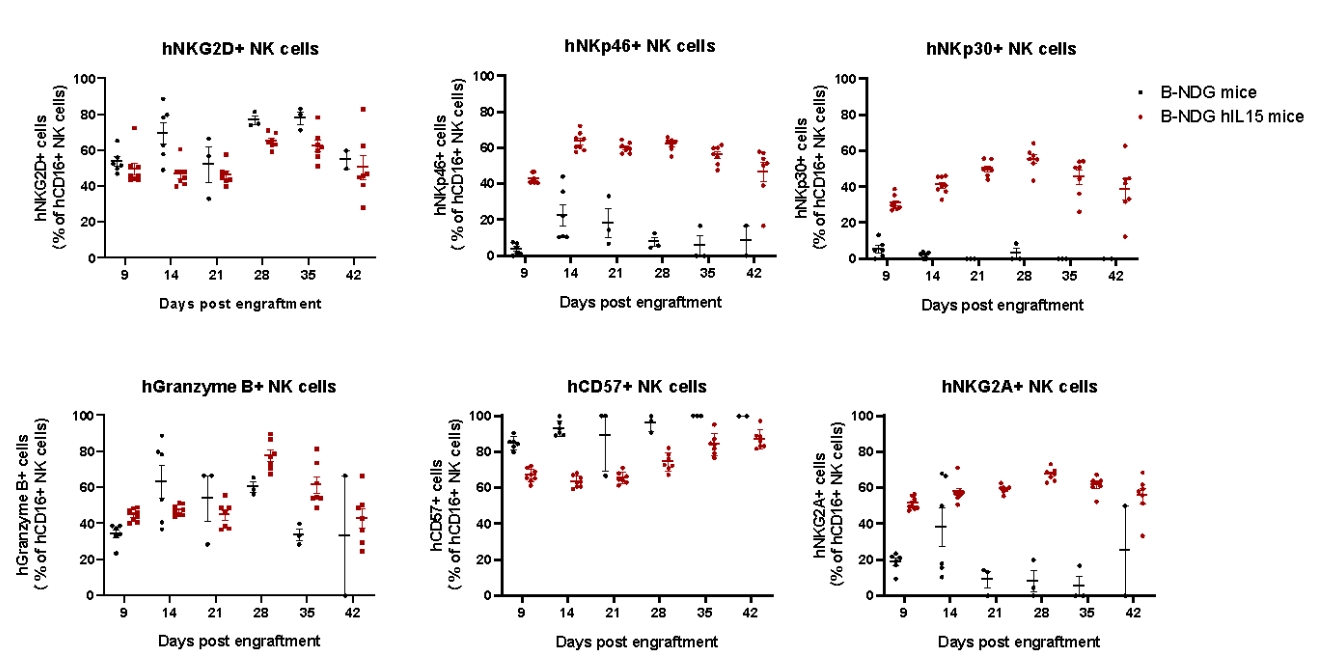
Expression analysis of functional markers and surface receptors in reconstituted human NK cells. NK cells were purified from human PBMC using the sorting kit, and sorted NK cells (2.0 M) were intravenously injected into B-NDG mice(female, 6 week-old, n=7)and B-NDG hIL15 mice(n=8)after treated with 1.2 Gy irradiation. Peripheral blood was collected weekly to detect cytotoxic markers Granzyme B and CD57, cell surface agonistic receptors NKG2D, NKp46 and NKp30, and the cell surface inhibitory receptor NKG2A in human NK cells. Result showed that compared with B-NDG mice, the proportions of hNKp46+ cells, hNKp30+ cells, hGranzyme B+ cells and human NKG2A+ cells in B-NDG hIL15 mice after human NK cell reconstitution were significantly increased, while the proportions of hNKG2D+ cells and hCD57+ cells were slightly decreased. These results indicate that the reconstituted human NK cells have cytotoxic activity.
-
In vivo efficacy of anti-human Claudin18.2 antibody with human NK cell engraftment

-
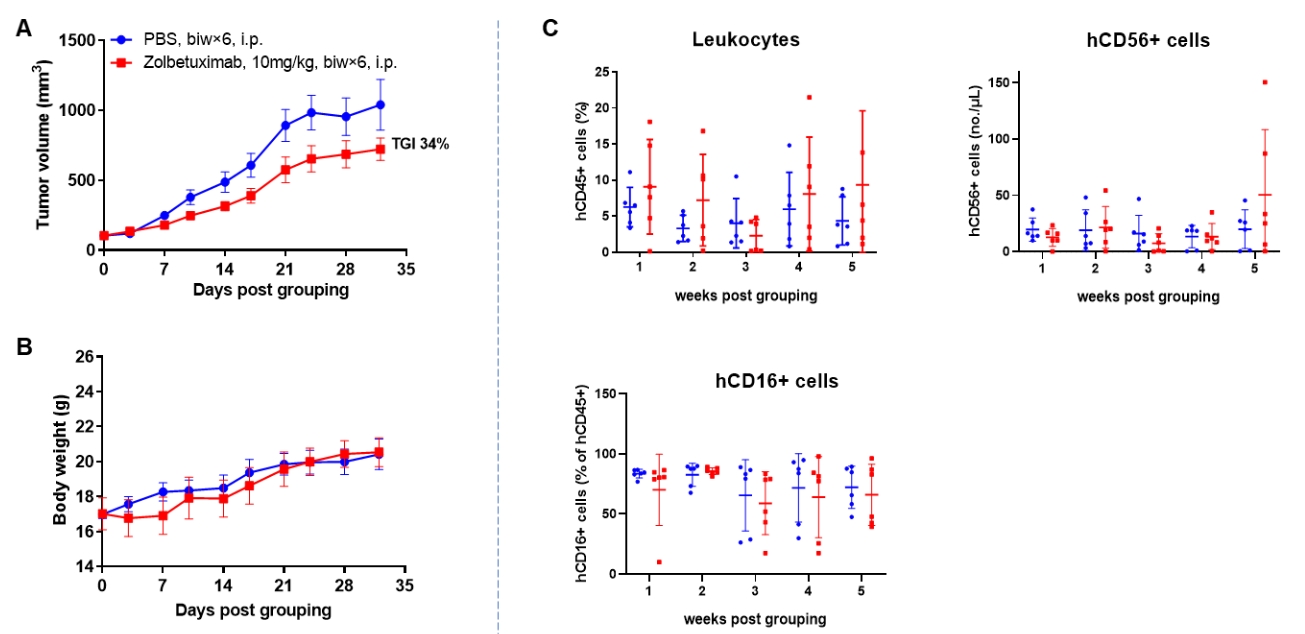
Antitumor activity of anti-human CLDN18.2 antibody in lung cancer cell line A549 CDX model established with huNK-B-NDG hIL15 mice. Human NK cells (2E6) purified from PBMCs were intravenously engrafted into B-NDG hIL15 mice irradiated with X-ray. B-hCLDN18.2 A549 cells (1E7) were subcutaneously inoculated into B-NDG hIL15 mice. The mice were grouped when the tumor volume reached approximately 100-150 mm3 at which time they was treated with anti-human CLDN18.2 antibody (zolbetuximab, in house) with dose and schedule indicated in panel (n=6). Peripheral blood was collected weekly to analyze the reconstitution levels of human CD45+cells and NK cells. The results showed that zolbetuximab could effectively inhibit tumor growth in huNK-B-NDG hIL15 mice. High levels of human NK cells could be detected in peripheral blood.
-
Human PBMC immune system engraftment

-
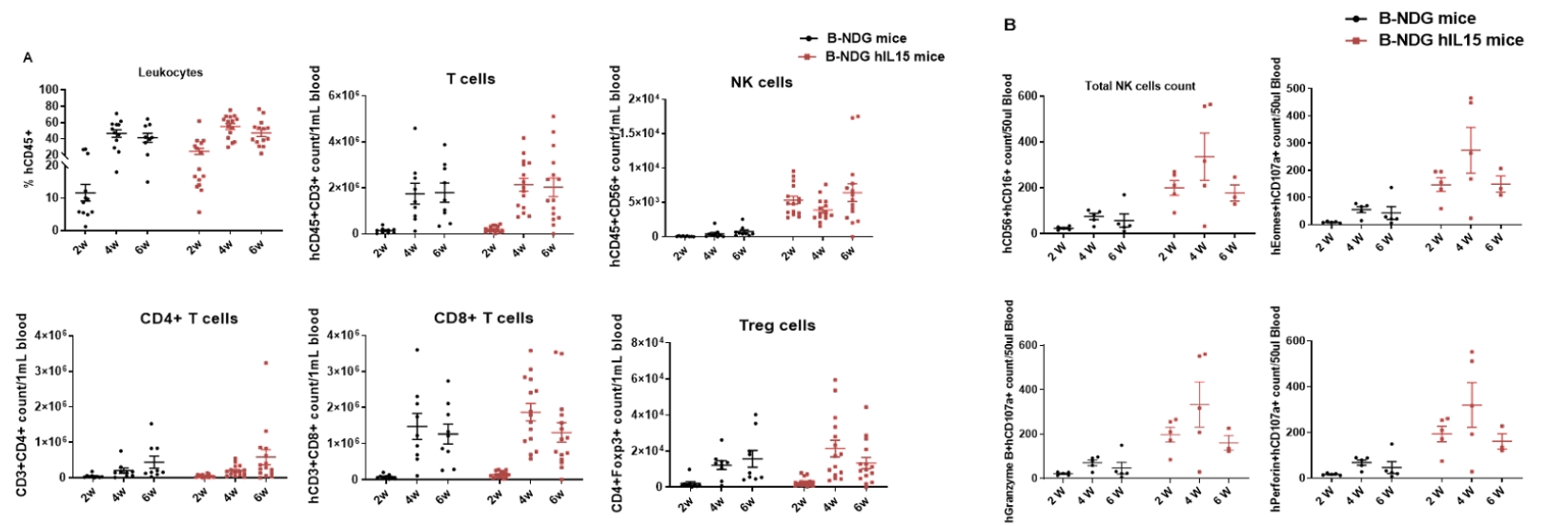
Human immune cell phenotyping in B-NDG hIL15 engrafted with human PBMC. Human PBMC cells (5E6) were intravenous implanted into homozygote B-NDG hIL15 (female, 5-week-old, n=15) and B-NDG mice (female, 5-week-old, n=10). Blood from B-NDG hIL15 and B-NDG mice was taken at different times after human PBMC engraftment for flow cytometric analysis. A. Human immune cell phenotyping. B. NK cell phenotyping. C. Survival. D. Body weight. Values were expressed mean ± SEM. Our results suggest that human NK and T cells in reconstituted B-NDG IL15 mice were successfully propagated.
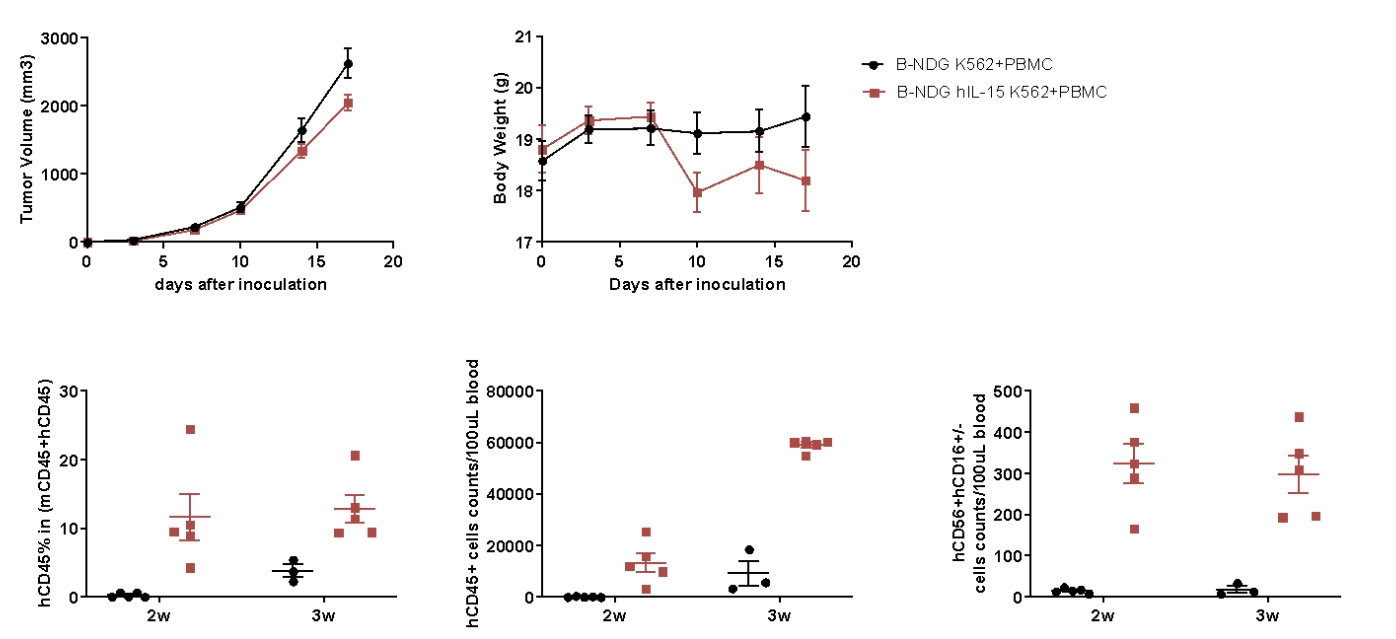
Analysis of CD45+ and NK cells after PBMC engraftment and K562 CDX model generation in B-NDG hIL15 mice . Human PBMC cells(5E6) were intravenous implanted into homozygote B-NDG hIL15 (female, 6 week-old, n=5) and B-NDG mice (female, 6 week-old, n=5). K562 cells(1E6) were subcutaneously implanted into B-NDG hIL15 and B-NDG mice on the same day as PBMCs. (A) The mean ± SEM of tumor sizes (B) The mean ± SEM of Body weight. (C) Blood from B-NDG hIL15 and B-NDG mice was taken at different times for flow cytometric analysis. K562 cells were successfully tumorigenic on a mouse model of human PBMC immune system reconstitution, and B-NDG hIL15 showed a higher percentage of human CD45+ cells and cell number of human NK cells compared to B-NDG.
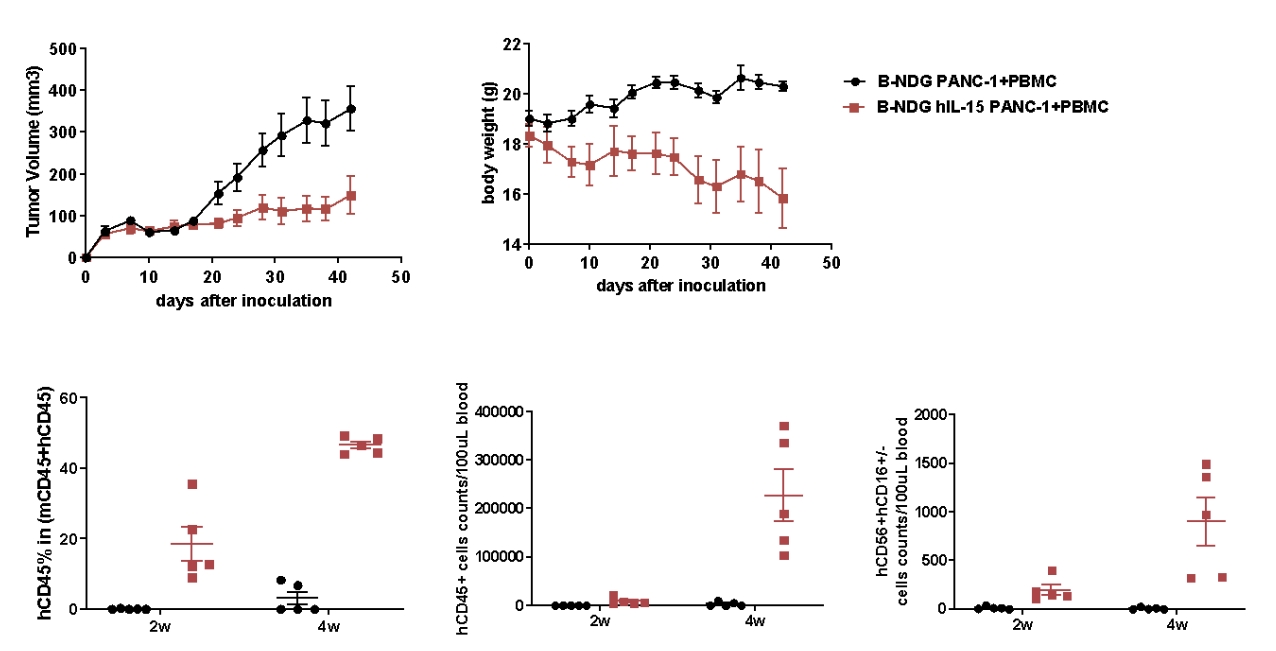
Analysis of CD45+ and NK cells after PBMC engraftment and PANC-1 CDX model generation in B-NDG hIL15 mice. Human PBMC cells(5E6) were intravenous implanted into homozygote B-NDG hIL15 (female, 6 week-old, n=5) and B-NDG mice (female, 6 week-old, n=5). PANC-1 cells(5E6) were subcutaneously implanted into B-NDG hIL15 and B-NDG mice on the same day as PBMCs. (A) The mean ± SEM of tumor sizes (B) The mean ± SEM of Body weight. (C)Blood from B-NDG hIL15 and B-NDG mice was taken at different times for flow cytometric analysis. PANC-1 cells were successfully tumorigenic on a mouse model of human PBMC immune system reconstitution, and tumor growth was significantly delayed in B-NDG hIL15 mice compared to B-NDG mice. B-NDG hIL15 showed a higher percentage of human CD45+ cells and cell number of human NK cells compared to B-NDG
-
In vivo efficacy of anti-human Claudin18.2 antibody with human PBMC engraftment

-
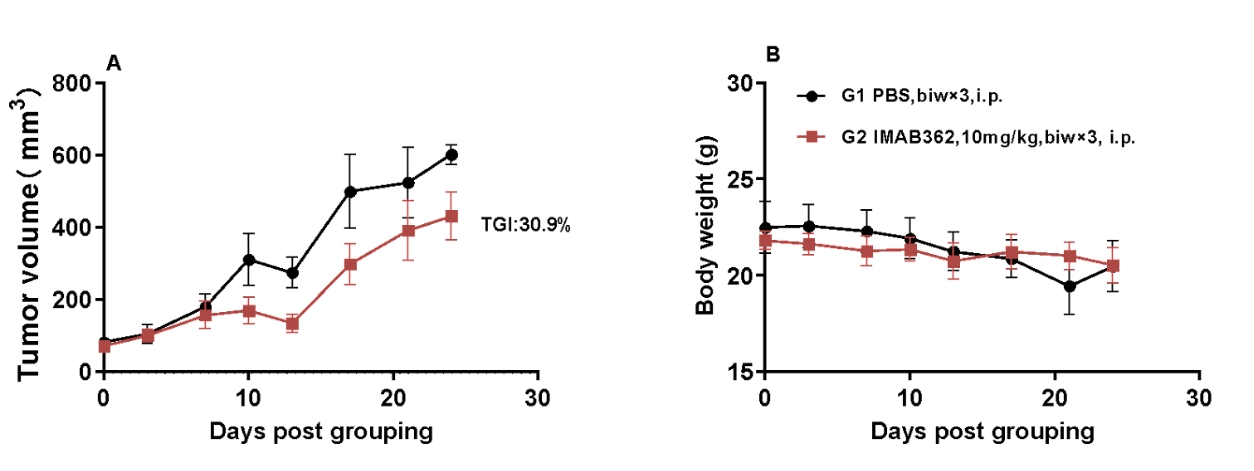
Antitumor activity of anti-human Claudin18.2 antibodies in B-NDG hIL15 mice. Human PBMC cells(1E6) were intravenous implanted into homozygote B-NDG hIL15 mice. NUGC4-CLDN18.2(6E6) were implanted into B-NDG IL15 mice. Mice were grouped when the tumor volume reached approximately 70-100mm3 (n=5) at which time they were treated with IMAB362 (in house) with doses and schedules indicated in panel. (A) Tumor volume changes during treatment, (B) Body weight changes during treatment. The results showed that CLDN18.2 antibodies had efficacies for tumor growth inhibition in B-NDG hIL15 mice with human PBMC immune system reconstitution model.
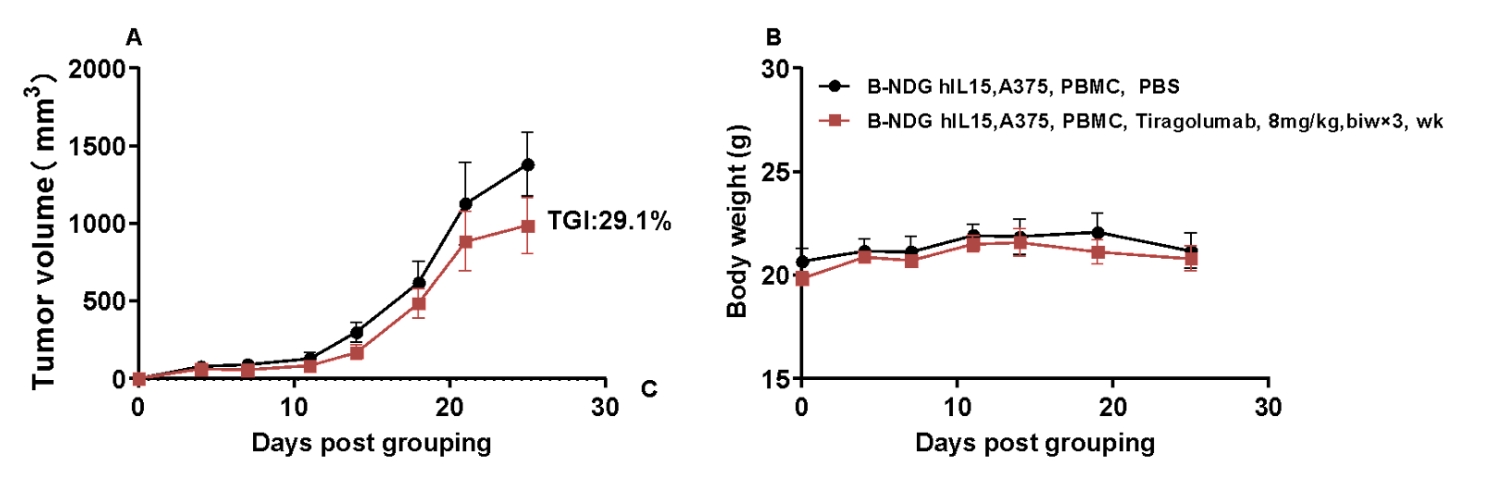
Antitumor activity of anti-human TIGIT antibodies in B-NDG hIL15 mice. Human PBMC cells(1E6) were intravenous implanted into homozygote B-NDG hIL15 mice. A375(4E6) were implanted into B-NDG hIL15 mice. Mice were grouped the day after tumor inoculation (n=5) and treated with Tiragolumab (in house) with doses and schedules indicated in panel. (A) Tumor volume changes during treatment, (B) Body weight changes during treatment. The results showed that TIGIT antibodies had efficacies for tumor growth inhibition in B-NDG hIL15 mice with human PBMC immune system reconstitution model.
-
Summary

-
- mRNA expression analysis:
Mouse Il15 mRNA was detectable only in splenocytes of wild-type B-NDG mice. Human IL15 mRNA was detectable only in homozygous B-NDG hIL15 mice, but not in B-NDG mice.
- Protein expression analysis:
Human IL15 was detectable in heterozygous and homozygous B-NDG hIL15 mice but not in B-NDG mice. Expression of human IL15 was stable during the passages of the mouse.
- Complete blood count (CBC) :
Peripheral blood of male and female B-NDG hIL15 mice (8 weeks old, n=10) was taken for complete blood count.
- Engraftment of human CD34+ HSCs or NK cells to reconstitute human immune system in B-NDG hIL15 mice:
- Engraftment of human CD34+ HSCs in adult and neonatal B-NDG hIL15 mice successfully reconstituted human T, B, NK cells and a small number of myeloid cells.
- Engraftment of human NK cells in adult B-NDG hIL15 mice successfully reconstituted functional NK cells.
- Establishment of CDX model with B-NDG hIL15 mice engrafted with human CD34+ HSCs:
Human tumor cell line, such as K562 and MV-4-11 were successfully tumorigenic in huHSC-B-NDG hIL15 mice.
- In vivo efficacy:
Anti-human Claudin18.2 antibody and anti-human EGFR antibody were efficacious in controlling tumor growth in huHSC-B-NDG hIL15 mice.
Anti-human Claudin18.2 antibody was efficacious in controlling tumor growth in huNK-B-NDG hIL15 mice.
-
Poster

-
B-NDG hIL-15 Mice Improve NK Cell Development After Human PBMC or CD34+ HSC Engraftment


