Basic Information
-
Gene targeting strategy

-
Gene targeting strategy of B-hPD-1/hTIM3 mice. The targeting strategy of B-hTIM3 mice is to the exon 2 of mouse Tim3 gene that encode the extracellular domain were replaced by human TIM3 exon 2 . The targeting strategy of B-hPD-1 mice is to the exon 2 of mouse PD-1 gene that encode the extracellular domain were replaced by human PD-1 exon 2. The B-hPD-1/hTIM3 double knock-in model, was developed by breeding the B-hPD-1 mice and the B-hTIM3 mice, has a functional mouse immune system.
-
mRNA expression analysis

-

Species-specific PD-1/TIM3 gene expression analysis in wild-type and humanized B-hPD-1/hTIM3 mice by RT-PCR. Mouse Pd-1/Tim3 mRNA was detected in splenocytes isolated from wild-type C57BL/6 (+/+) mice, while human PD-1/TIM3 mRNA was detected in B-hPD-1/hTIM3 mice.
-
Protein expression analysis

-
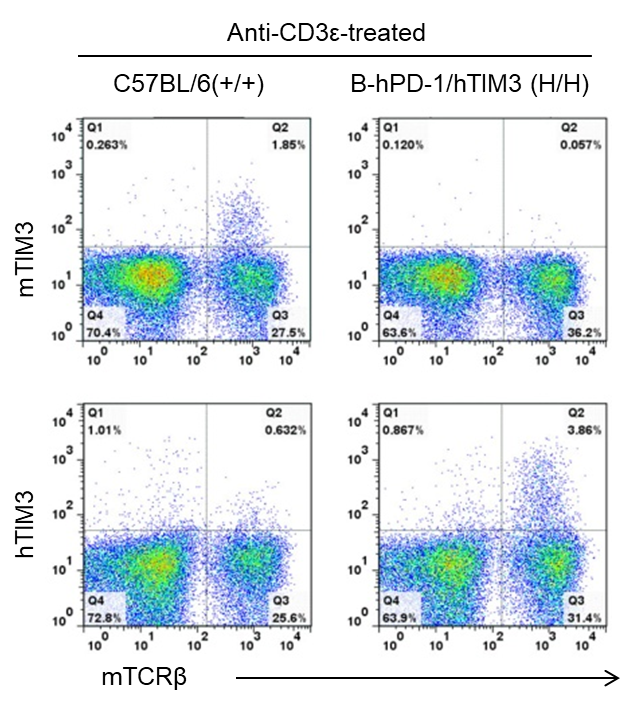
Species-specific TIM3 protein expression analysis in wild-type and humanized B-hPD-1/hTIM3 mice. Splenocytes were isolated from wild-type C57BL/6 (+/+) and homozygous B-hPD-1/hTIM3 (H/H) mice stimulated with anti-CD3ε in vivo, and analyzed by flow cytometry using species-specific anti-TIM3 antibodies. Mouse TIM3 protein was detected in wild-type mice, while human TIM3 protein was detected in B-hPD-1/hTIM3 mice.
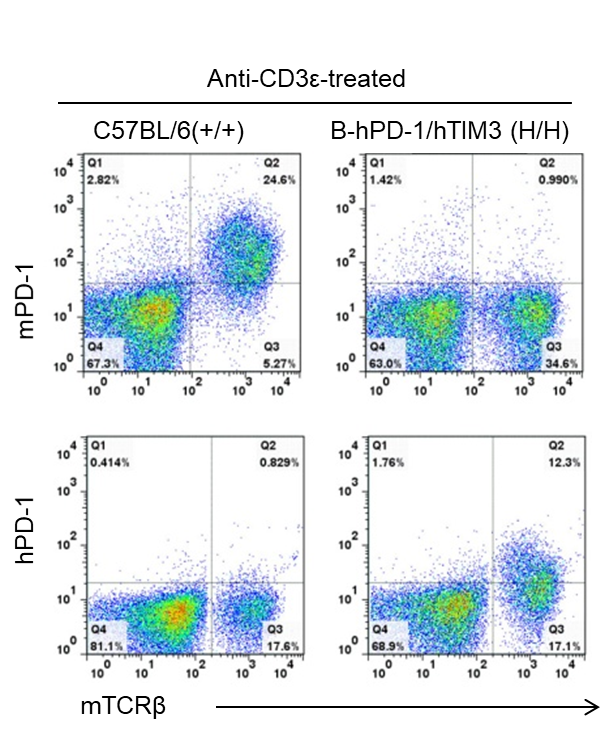
Species-specific PD-1 protein expression analysis in wild-type and humanized B-hPD-1/hTIM3 mice. Splenocytes were isolated from wild-type C57BL/6 (+/+) and homozygous B-hPD-1/hTIM3 (H/H) mice stimulated with anti-CD3ε in vivo, and analyzed by flow cytometry using species-specific anti-PD-1 antibodies. Mouse PD-1 protein was detected in wild-type mice, while human PD-1 protein was detected in B-hPD-1/hTIM3 mice.
-
Combination therapy

-
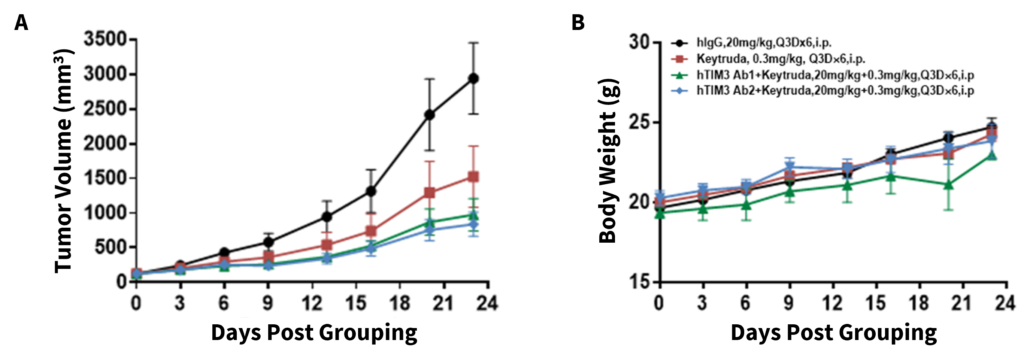
Antitumor activity of anti-human TIM3 and anti-human PD-1 antibodies in B-hPD-1/hTIM3 mice. Murine colon cancer MC38 cells (5×105) were subcutaneously implanted into homozygous B-hPD-1/hTIM3 mice (female, 6-8 week-old, n=6). Mice were grouped when tumor volume reached approximately 150±50 mm3, at which time they were treated with anti-human TIM3 and/or anti-human PD-1 (Keytruda) antibodies at doses and schedules as indicated. (A) Anti-human TIM3 antibodies (Ab1/Ab2) combined with Keytruda inhibited MC38 tumor growth in B-hPD-1/hTIM3 mice, (B) without negatively impacting body weight changes. Values are expressed as mean ± SEM.
Antitumor activity of anti-human TIM3 and anti-human PD-1 antibodies in B-hPD-1/hTIM3 mice. (A) Anti-hTIM3 antibodies (provided by a client) combined with anti-hPD-1 antibodies (Keytruda) inhibited MC38 tumor growth in B-hPD-1/hTIM3 mice. Murine colon cancer MC38 cells (5×105) were subcutaneously implanted into homozygous B-hPD-1/hTIM3 mice (female, 6-8 week-old, n=6). Mice were grouped when tumor volume reached approximately 150±50 mm3, at which time they were treated with anti-hTIM3 antibodies and anti-hPD-1 antibodies (Keytruda) with doses and schedules indicated in the panel. (B) Body weight changes during treatment. As shown in panel A, the combination of anti-hTIM3 Ab2 and anti-hPD-1 antibody treatment shows enhanced tumor growth inhibition compared to individual groups, demonstrating that the B-hPD-1/hTIM3 mice provide a powerful preclinical model for in vivo evaluating combination therapy efficacy of hTIM3 antibodies and hPD-1 antibodies. Values are expressed as mean ± SEM.
-
Related products

-
-
References

-
Nat Commun. 2017 Feb 6;8:14369. doi: 10.1038/ncomms14369.
EMBO J. 1992 Nov;11(11):3887-95.
Int. J. Mol. Sci. 2017, 18, 645; doi:10.3390/ijms18030645


